Ethanol Shelf Life & Expiration: Storage and Stability Guide
Ethanol is a versatile organic solvent used across food manufacturing, pharmaceuticals, cosmetics, laboratories, and industrial processes. Knowing its shelf life and storage requirements helps you keep product quality and effectiveness throughout its useful period. While exceptionally stable compared to many organic compounds, ethanol needs attention to storage conditions to prevent evaporation, contamination, and concentration changes.
Ethanol stability depends on purity, storage environment, and packaging. High-purity solutions last remarkably long under appropriate conditions, while exposure to air, light, and heat can reduce quality. This article offers thorough guidance on extending ethanol shelf life and identifying when replacement is needed.


In this article, we’ll review:
- How Long Does Ethanol Last?
- Signs That Ethanol Has Expired
- What to Do If Ethanol Is Expired
How Long Does Ethanol Last?
Ethanol longevity depends on several factors, including grade, concentration, packaging, and storage environment. Knowing these variables helps you estimate how long it will remain effective and plan storage strategies.
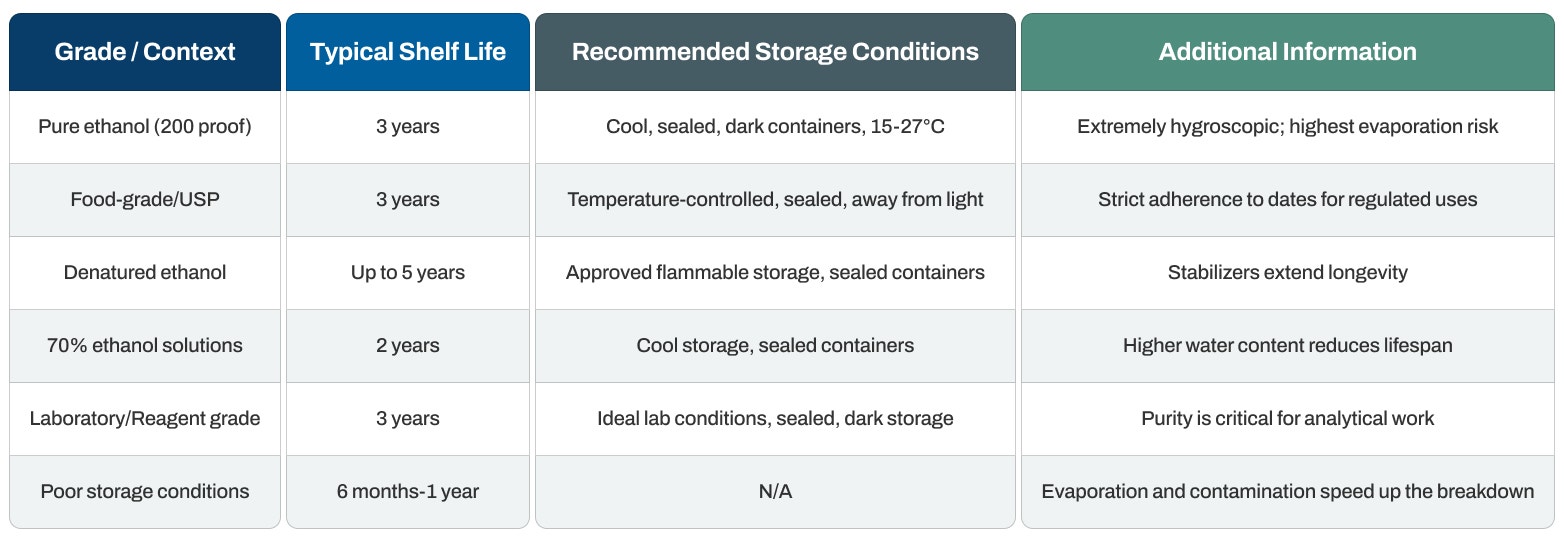
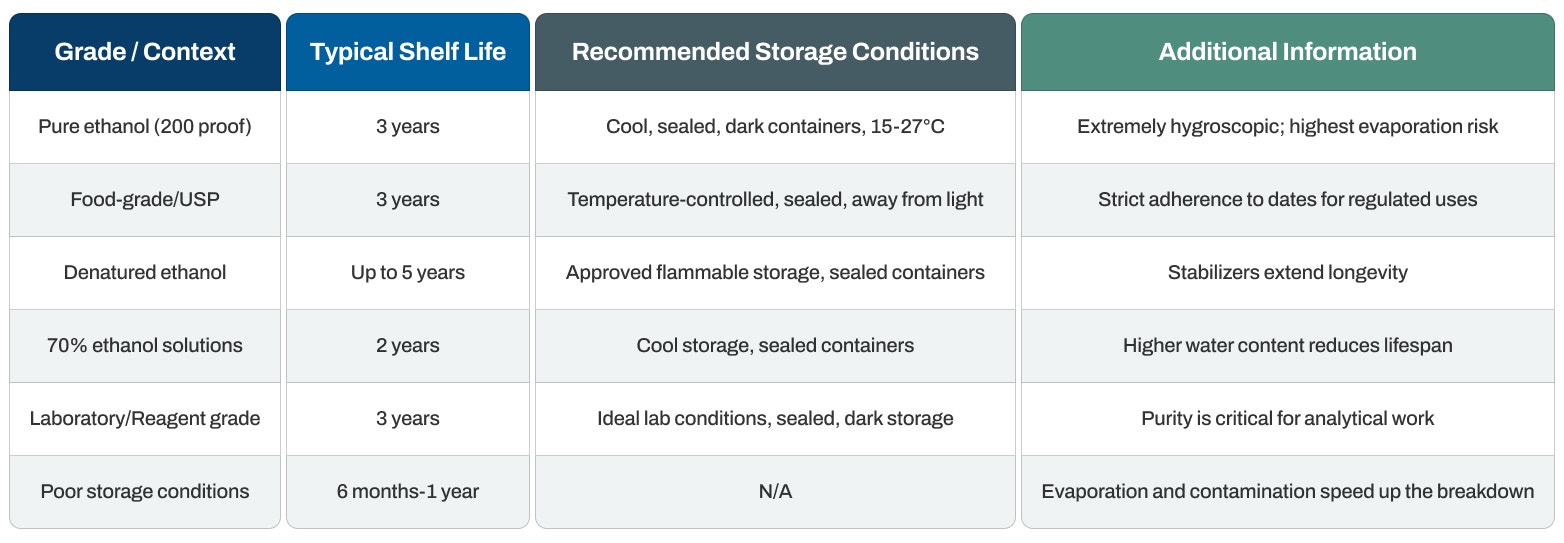
Shelf Life Duration
Pure ethanol (200 proof): Typically remains effective for 3 years from the manufacturer's date when stored in ideal conditions. The absence of water contributes to better stability.
Food-grade and pharmaceutical (USP) ethanol: It retains its 3-year lifespan from manufacture when stored correctly in sealed containers. High purity standards and careful packaging support longer storage.
Denatured ethanol formulations: Can last up to 5 years because added stabilizers and denaturants boost stability. The particular denaturant formula influences how long it lasts.
70% ethanol solutions: Have a briefer lifespan of roughly 2 years because increased water content creates a slightly higher risk of microbial contamination in poorly sealed containers.
Optimal storage potential: When kept correctly in sealed containers away from heat and light, pure ethanol can theoretically remain usable indefinitely because of its excellent chemical stability.
Chemical Stability
Ethanol solutions show exceptional stability under normal conditions, though several factors affect quality retention:
- Baseline stability: The compound doesn't readily break down under normal storage conditions, allowing long-term inventory management when environmental controls work well. Ethanol actually functions as a natural antimicrobial agent, stopping microbial growth and contamination in sealed containers.
- Degradation potential: Can theoretically break down to form acetaldehyde under extreme conditions with prolonged heat and light exposure, but this rarely happens during normal storage. The main concern is evaporation rather than chemical breakdown.
- Temperature sensitivity: Ethanol has a boiling point of 78.37°C (173°F), making evaporation the chief quality issue rather than thermal breakdown. Even at room temperature, poor sealing allows gradual evaporation that raises concentration.
- Light-induced effects: UV light exposure can cause pure ethanol to fail within 3 months, depending on packaging. Photochemical reactions may create trace amounts of breakdown products that affect quality in sensitive analytical applications.
- Hygroscopic nature: Ethanol readily pulls moisture from the air, particularly in absolute ethanol grades. This moisture uptake dilutes concentration and impacts performance in applications needing precise alcohol content.
Purity & Packaging
Packaging decisions greatly affect how long ethanol keeps its quality:
- Purity advantages: Higher purity grades (190-200 proof) in sealed containers stay good longer than diluted solutions. The lower water content reduces opportunities for microbial growth and chemical interactions.
- Container materials: Non-reactive materials like HDPE plastic, glass, and stainless steel offer the best protection for long-term storage. These materials resist corrosion and don't release contaminants into ethanol.
- Reactive materials: Steer clear of untreated aluminum or certain brass alloys that may corrode or release contaminants over time. Some plastics may break down or become permeable when exposed to ethanol long-term.
- Original packaging: Sealed containers from manufacturers offer the best protection against evaporation and contamination. These containers are specifically designed to keep seal integrity and prevent concentration drift.
- Seal integrity: Correct sealing stops evaporation and concentration changes that modify product specifications. Even small leaks allow alcohol vapor to escape and atmospheric moisture entry, impacting quality over time.
Storage Conditions
Environmental management creates the foundation for successful long-term ethanol storage:
- Temperature range: Best kept in a cool, dry, well-ventilated area at 15-27°C (59-80°F) or 10-25°C (50-77°F) for best stability. This range stops excessive evaporation while preventing any potential microbial growth in diluted solutions.
- Light protection: Shield from direct sunlight and UV light exposure through amber containers or storage in dark areas. Photochemical breakdown proceeds slowly but builds up over extended storage periods, particularly affecting high-purity analytical grades.
- Humidity control: Keep relative humidity under 50% to prevent moisture uptake, as ethanol is hygroscopic. High humidity raises the risk of water contamination, particularly when containers are opened frequently.
- Fire safety: Keep away from ignition sources, sparks, and electrical equipment because of high flammability. Apply approved flammable liquid storage cabinets that offer fire protection and correct ventilation.
- Sealing protocol: Make sure containers stay tightly sealed to stop evaporation and concentration changes. Regular inspection verifies seal integrity throughout the storage period, particularly important for high-purity grades.
Quick Reference: Shelf Life and Storage
A quick and appropriate first aid response can significantly reduce the severity of ethanol exposure injuries. Immediate action remains the priority for all exposure routes.
Signs That Ethanol Has Expired
Physical and performance changes show when ethanol has broken down beyond acceptable quality levels. Early recognition stops the use of compromised material that could impact your processes or products.
- Concentration Changes: Major deviation from labeled proof or percentage because of evaporation, impacting effectiveness in analytical, extraction, or formulation applications. Even minor concentration changes affect the results in precision work.
- Moisture Contamination: Evidence of water uptake resulting in lower alcohol content, particularly noticeable in high-purity absolute ethanol kept in high-humidity conditions. Cloudiness may appear in previously clear solutions.
- Discoloration: Appearance of yellow or amber color in normally clear ethanol, showing breakdown or contamination from packaging materials or environmental exposure. Fresh ethanol should be water-clear.
- Unusual Odors: Off-smells or foreign odors that weren't there in fresh ethanol, suggesting contamination from storage containers, environmental pollutants, or breakdown products. Pure ethanol has a characteristic, clean, slightly sweet odor.
- Sediment or Particulates: Appearance of visible particles, cloudiness, or foreign matter in the solution. This may show contamination from packaging materials, environmental sources, or chemical reactions with container materials.
- Container Integrity Issues: Cracks, leaks, or broken seals that compromise the ethanol's purity and concentration. Damaged containers allow evaporation and atmospheric contamination.
- Performance Issues: Decreased effectiveness in intended applications like extraction, dissolution, disinfection, or analytical procedures. Concentration changes directly affect performance in precision applications.


What to Do If Ethanol Is Expired
Finding expired ethanol needs a systematic response to protect product quality, ensure safety, and keep regulatory compliance across your applications.
Check Manufacturer Guidelines
Review packaging labels and certificates of analysis for expiry or retest dates. Food-grade and pharmaceutical applications require strict adherence to expiration dates for safety and regulatory compliance. These applications typically forbid the use of expired material regardless of apparent condition.
Laboratory and industrial applications may permit retesting by qualified personnel to decide continued suitability, particularly for non-critical uses. Yet analytical work needing precise concentrations should always use fresh, certified material.
Follow Proper Disposal Procedures
Dispose of expired ethanol in accordance with local hazardous waste regulations. Ethanol is classified as flammable and must be handled as hazardous waste. Never pour down drains or throw away in regular trash, as this can create fire hazards and violate environmental regulations.
Small quantities may be allowed for sink disposal in some jurisdictions if heavily diluted, but check local regulations first. Large quantities need to be collected by licensed hazardous waste handlers. Mark disposal containers clearly as "Flammable Liquid Waste" and keep in approved areas away from ignition sources until pickup.
Work in well-ventilated areas when handling expired ethanol for disposal. Remove all ignition sources and use suitable personal protective equipment, including chemical-resistant gloves and safety goggles.
Replace with Fresh Stock
Get replacement ethanol from reputable suppliers offering thorough documentation, including certificates of analysis with current manufacture dates. Check grade and purity specifications match your application needs, whether for pharmaceutical formulations, laboratory analysis, food extraction, or industrial processes.
Look over incoming shipments for packaging integrity, correct sealing, and clear storage recommendations. Verify concentration specifications align with your process needs before accepting delivery. Check that containers show no signs of damage or leakage during transport.
Keep in Mind: Proper Storage Extends Shelf Life
Put in place strong storage protocols from the start to extend lifespan and keep quality:
- Container selection: Apply suitable containers built from HDPE, glass, or stainless steel that resist ethanol and prevent vapor permeation. Original manufacturer packaging often offers the best protection until first use.
- Temperature control: Keep storage areas at 15-27°C while protecting from heat sources. Install thermometers in storage areas to watch conditions continuously.
- Light protection: Keep in dark areas or use amber containers to reduce photochemical breakdown. Place containers in closed cabinets or storage rooms without windows when possible.
- Seal maintenance: Make sure to tighten the sealing after each use to stop evaporation and concentration drift. Apply containers with positive-seal caps rather than simple screw caps for long-term storage.
- Fire safety compliance: Keep in approved flammable liquid storage cabinets with correct ventilation. Keep adequate clearance from ignition sources and post suitable hazard warnings.
- Inventory rotation: Apply older stock first through systematic inventory management, stopping the accumulation of aging material. Date containers upon receipt and mark with "opened" dates when first used.
- Environmental monitoring: Regular checks of storage conditions spot problems needing adjustment before product quality suffers. Address temperature excursions, humidity issues, or ventilation problems quickly.
Conclusion
Ethanol generally lasts 3 years from manufacture for pure grades with excellent stability, though it can stay viable indefinitely under correct storage conditions. Denatured formulations may stretch to 5 years, while diluted solutions like 70% ethanol have briefer lifespans of roughly 2 years. The main concerns are evaporation, causing concentration changes, and light-induced breakdown. Correct storage in sealed, non-reactive containers away from heat, light, and ignition sources is vital for keeping quality and extending the lifespan of this highly stable and versatile solvent.
Note: Fresh ethanol kept under ideal conditions delivers reliable performance throughout its lifespan when handled according to safety guidelines and manufacturer specifications for your particular application.
For more detailed guidance, explore our Resource Library for comprehensive storage and handling information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Ethanol You Can Trust?
If you are looking for high-quality ethanol for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's ethanol is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.











