About Isooctane
Isooctane is less ambiguously known as 2,2,4-trimethylpetane, and belongs to a group of several isomers that share the formula C8H18. It is a colorless liquid with a petroleum-like odor. Isooctane is mass-produced by the petroleum industry via the catalyzed reaction of isobutene with isobutane.
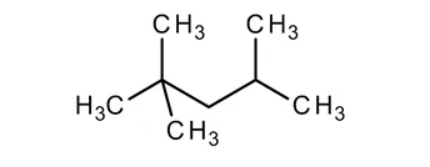
Isooctane is principally used as an octane booster in fuel, but also as a standard to determine the octane rating of fuel. As a fuel additive, isooctane is known to prevent “engine knocking.” This property later became the basis of the octane rating scale, in which the performance of isooctane is given the rating of 100 and that of heptane is standardized to 0.
In instrumental analysis, isooctane is considered a secondary reference standard, which can be employed for method verification practices and instrument calibration. It also finds use in organic synthesis.
Major uses and applications
The following areas are the most prevalent applications for isooctane.
- Petroleum industry: Isooctane is principally used as an octane booster in fuel and as a standard to determine octane rating.
- Chemical synthesis: Isooctane may be used as a reagent in organic chemical synthesis.
- Instrumental analysis and pharmaceutical standards: Isooctane is a secondary reference standard, used for method verification and instrument calibration.
Common Uses and Applications
- Octane booster in fuel
- Fuel standard
- Analytical standard
- Laboratory solvent
- Organic chemical synthesis
Industries
- Petroleum industry
- Pharmaceutical industry







