Caffeine Safety & Hazards: Essential Handling Guidelines
Caffeine is a white, odorless, crystalline powder or glistening needles with a bitter taste, naturally occurring in coffee, tea, and cocoa, and widely used in pharmaceuticals, beverages, dietary supplements, and cosmetics. Caffeine is classified as harmful if swallowed with moderate acute toxicity (oral LD50 192 mg/kg in rats) and acts as a central nervous system stimulant, causing dose-dependent effects from alertness to serious toxicity, including seizures and cardiac arrhythmias. While caffeine is not flammable and presents low fire risk, it can form combustible dust clouds, and its primary hazards involve acute oral toxicity, CNS stimulation, cardiovascular effects, and potential reproductive concerns. Proper handling requires dust control, appropriate PPE, and awareness of toxicity symptoms.
Professionals working with bulk caffeine in pharmaceutical manufacturing, dietary supplement production, beverage formulation, and chemical laboratories face different hazards than consumers using finished products. Understanding the toxicological profile of concentrated caffeine helps prevent accidental poisoning and ensures safe manufacturing processes.
Pure caffeine powder poses serious ingestion risks that don't exist with beverages or finished products. The estimated lethal dose for an average adult is approximately 10-14 grams, an amount that looks deceptively small as a white powder. Children face even greater risk with toxic effects occurring at much lower doses. Beyond toxicity concerns, caffeine dust can irritate respiratory passages and eyes, while fine particles may form combustible clouds under specific conditions. This guide provides essential safety information for handling concentrated caffeine in industrial and laboratory settings.


In this article, we’ll explore:
-
Chemical Hazards Overview
-
Exposure Risks & Health Effects
-
First Aid Measures
-
Handling & Storage Precautions
-
Spill & Leak Procedures
-
Fire & Explosion Hazards
-
Regulatory & Compliance Information
-
Pharmacological & Toxicological Profile
-
Technical Documents & References
Chemical Hazards Overview
Caffeine's hazard profile centers on its pharmacological activity as a central nervous system stimulant rather than chemical reactivity or flammability.
Hazard Classification
Under the Globally Harmonized System, caffeine receives an exclamation mark pictogram (⚠️ GHS07) for acute toxicity and harmful effects. The material is classified as harmful if swallowed (Acute Toxicity Category 4, H302). It may cause eye, skin, and respiratory tract irritation along with digestive tract irritation. The compound may trigger cardiac disturbances and central nervous system effects. Dust may present a combustible dust hazard under specific conditions.
NFPA hazard ratings place caffeine at Health: 1-2 (slight to moderate hazard), Flammability: 1 (slight), and Reactivity: 0 (stable).
Primary Risks
Toxicity data show oral LD50 values of approximately 192 mg/kg in rats, 127 mg/kg in mice, and 224 mg/kg in rabbits. These animal studies translate to an estimated human lethal dose of around 192 mg/kg, or roughly 10-14 grams for an average adult. The material is harmful if swallowed and causes central nervous system stimulation at lower doses.
Cardiovascular effects may occur with excessive exposure. Dust may cause mechanical irritation to the skin, eyes, and respiratory passages. Under certain conditions, fine caffeine dust can form combustible dust clouds, though this represents a lower risk than with many organic powders.
Exposure Risks & Health Effects
Recognizing how caffeine affects the body at different exposure levels helps you identify when dangerous situations are developing.
Inhaling dust may irritate your respiratory tract, causing coughing and throat discomfort. The dust may also cause central nervous system stimulation effects if inhaled in significant quantities. Risk remains low under normal handling because caffeine has negligible vapor pressure at room temperature and doesn't readily become airborne unless you disturb the powder. Adequate ventilation minimizes dust exposure during transfers and weighing operations.
Skin contact may cause mild irritation with prolonged exposure. Animal studies show dermal LD50 values exceeding 2000 mg/kg in rats, indicating low toxicity through skin absorption. Dust contacting skin causes mechanical irritation but isn't readily absorbed through intact skin. Washing thoroughly after handling removes residual powder.
Getting dust in your eyes causes mechanical irritation with redness, tearing, and discomfort. Serious or permanent eye damage is not expected. The irritation typically resolves after thorough rinsing removes particulates.
Swallowing caffeine powder is the most serious exposure route. Animal toxicity studies show oral LD50 values of 192 mg/kg in rats, 127 mg/kg in mice, and 224 mg/kg in rabbits. The human oral lethal dose is estimated at approximately 192 mg/kg, translating to 10-14 grams for an average adult. Children are particularly vulnerable, with doses as low as 140 mg/kg reported as toxic.
Acute poisoning symptoms include nausea, vomiting, and diarrhea. Central nervous system stimulation manifests as nervousness, restlessness, insomnia, and tremors. Cardiovascular effects include rapid heartbeat (tachycardia) and irregular heart rhythms (arrhythmias). Gastrointestinal distress accompanies most overdoses. Severe poisoning may progress to seizures, delirium, respiratory failure, and death. Chronic excessive use causes digestive disturbances, heart palpitations, anxiety, and sleep disorders.
Repeated exposure over time may affect the heart, gastrointestinal tract, and central nervous system. Caffeine is not classified as a carcinogen by IARC (Category 3), ACGIH, NIOSH, NTP, or OSHA. Laboratory studies show mutagenic activity in mammalian somatic cells and bacteria/yeast cultures. The material may cause adverse reproductive effects and has been reported as a human teratogen, causing developmental abnormalities of the craniofacial and musculoskeletal systems. High doses may cause pregnancy termination and stillbirth. Excessive use can lead to caffeine dependence with withdrawal symptoms upon cessation.
First Aid Measures
Responding quickly to caffeine exposure prevents progression from minor irritation to serious toxicity, particularly with ingestion cases.
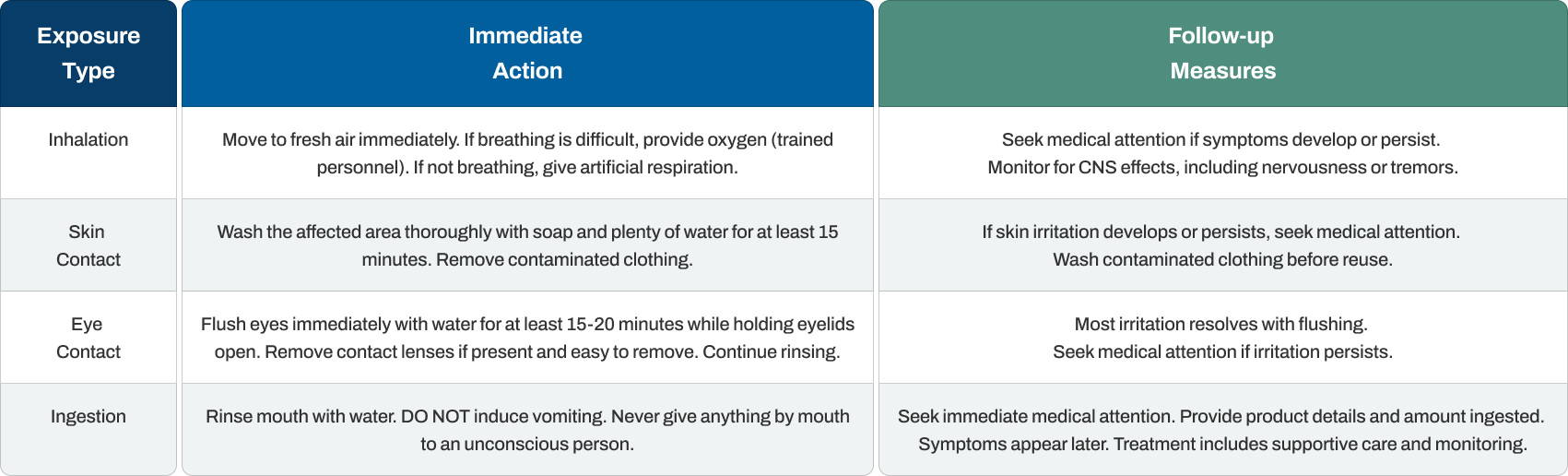
Caffeine poisoning symptoms may take several hours to fully develop, making medical observation important even if the person seems fine initially. Treatment focuses on supportive care since no specific antidote exists. Healthcare providers may administer activated charcoal to reduce absorption, perform gastric lavage for recent large ingestions, provide intravenous fluids, and monitor cardiac function for arrhythmias.
Handling & Storage Precautions
Safe caffeine handling emphasizes dust control and preventing accidental ingestion while recognizing the material's low chemical reactivity.
Safe Handling Procedures
Avoid creating dust during transfers, weighing, and processing operations. Work in well-ventilated areas or use local exhaust ventilation to capture dust at the source. Wear appropriate gloves, safety goggles, and protective clothing to prevent skin and eye contact with powder.
Never eat, drink, or smoke while handling caffeine. This prevents accidental ingestion and cross-contamination of food or beverages. Wash hands thoroughly after handling and before breaks or the end of the shift. Use non-sparking tools when handling bulk quantities to avoid potential dust ignition. Ensure proper grounding to prevent static buildup with fine powders.
Storage Conditions
Store in cool, dry, well-ventilated areas away from moisture and physical damage. Keep containers tightly closed when not in use to prevent moisture absorption and contamination. Protect from freezing and temperature extremes. Store away from food, beverages, and animal feed to prevent cross-contamination.
Maintain storage temperature below 25°C (77°F) for optimal stability. Keep away from incompatible materials, though caffeine is generally stable and not highly reactive. Store locked up if required by local regulations, particularly in facilities where unauthorized access could lead to misuse.
Incompatible Substances
Keep caffeine away from strong oxidizing agents. The material is generally stable and not highly reactive with most common chemicals. Avoid conditions that generate dust clouds near ignition sources, as fine organic dust can pose combustion hazards.
Spill & Leak Procedures
Caffeine spills present minimal fire hazard but require dust control to prevent inhalation exposure and potential combustible dust formation.
Emergency Response Steps
Avoid creating dust clouds during cleanup. Ensure adequate ventilation in the spill area. Wear appropriate personal protective equipment, including a dust respirator, gloves, safety goggles, and protective clothing. Eliminate ignition sources if significant dust generation is possible, though fire risk remains low. Restrict access to prevent tracking contamination and unauthorized exposure.
Containment Methods
For small spills, use appropriate tools or a vacuum to collect spilled powder into a waste disposal container. For large spills, use non-sparking tools and equipment as a precaution. Avoid sweeping, which creates dust clouds and increases inhalation exposure.
Use HEPA-filtered vacuums when available to capture fine particles effectively. Prevent entry into drains, sewers, or waterways where the material could contaminate water supplies.
Proper Disposal
Collect spilled material for proper disposal as potentially hazardous waste. Contact a licensed waste disposal service familiar with pharmaceutical and chemical waste handling.
The material may be dissolved or mixed with combustible solvent and burned in an approved chemical incinerator equipped with afterburner and scrubber systems. Dispose according to federal, state, and local regulations. Never dispose of bulk caffeine with household garbage due to toxicity concerns.
Fire & Explosion Hazards
Caffeine presents low fire risk under normal conditions but requires awareness of combustible dust potential with fine powders.
Flammability
Caffeine is not classified as a flammable liquid. Flash point testing is not applicable, or results exceed 93°C (200°F) in closed cup methods. The melting point ranges from 234-237°C (453-459°F), well above normal handling temperatures. Under normal conditions, the material is not flammable.
Caffeine becomes slightly flammable to flammable in the presence of open flames, sparks, or heat sources. The primary fire concern involves the dust explosion hazard. Fine caffeine powder may form combustible dust clouds in the air under certain conditions, particularly during bulk transfers, grinding operations, or if dispersed by compressed air.
Extinguishing Media
Use water spray, fog, or foam for caffeine fires. Dry chemical powder and carbon dioxide (CO2) work effectively. Alcohol-resistant foam provides good coverage for larger fires. For small fires, dry chemical powder offers a quick knockdown.
For large fires involving significant quantities, water spray or foam works best. Avoid using water jets, which may spread fire and create steam hazards.
Firefighting Precautions
Firefighters should wear self-contained breathing apparatus and full protective gear. The product is toxic, so avoid inhaling fire gases or vapors. Cool fire-exposed containers with water spray to prevent rupture. Combustion produces toxic gases, including carbon monoxide, carbon dioxide, and nitrogen oxides.
Evacuate areas as needed based on fire size and toxic gas generation. Be aware of dust explosion potential if fine powder becomes dispersed in enclosed spaces during fire conditions.
Regulatory & Compliance Information
Caffeine faces regulation as both a pharmaceutical ingredient and a toxic substance, depending on use and concentration.
OSHA Regulations
1910.1200). No established OSHA Permissible Exposure Limit (PEL) exists specifically for caffeine. The material is not designated as a highly hazardous substance under specific OSHA standards. Employers should follow general industry hygiene and safety practices, including dust control, appropriate PPE, and worker training on toxicity hazards.
EPA & Environmental Impact
Caffeine is not listed under CERCLA as a hazardous substance, so no reportable quantity (RQ) applies to environmental releases. The material is not subject to SARA Title III Section 302 (Extremely Hazardous Substances) or Section 313 (Toxics Release Inventory) reporting requirements.
In environmental systems, caffeine is readily biodegradable and is expected to break down rather than bioconcentrate in aquatic organisms. The compound shows high soil mobility and is not expected to volatilize from soil or water surfaces. Atmospheric half-life is approximately 2.5 hours in the particulate phase. Caffeine is not classified as persistent or bioaccumulative in the environment.
DOT Transportation Regulations
Caffeine falls under Hazard Class 6.1 (Poisonous/Toxic Material) for transportation. The proper shipping name is "Alkaloids, solid, n.o.s. (Caffeine)." The UN number is UN1544. Packing Group III classification reflects moderate toxicity. Shipments require toxic substance labels and comply with DOT shipping regulations for toxic materials.
FDA/USP Compliance
The FDA approves caffeine as an active pharmaceutical ingredient and food additive. USP/NF-grade caffeine is available and meets pharmaceutical compendial standards for use in drug manufacturing. The material appears in FDA-approved drug products, including pain relievers, cold medications, and stimulants.
Caffeine has a Generally Recognized As Safe (GRAS) status for certain food and beverage applications at appropriate levels. The FDA establishes maximum levels for caffeine in foods and beverages. European Union classification lists caffeine as harmful if swallowed (R22), possible risk of irreversible effects (R40), and possible risk of harm to the unborn child (R63).
Pharmacological & Toxicological Profile
Understanding caffeine's mechanism of action and dose-response relationship helps you recognize toxicity symptoms and implement appropriate safety measures.
Mechanism of Action
Caffeine functions as a central nervous system stimulant, acting primarily as an adenosine receptor antagonist. Blocking adenosine receptors, it increases alertness, reduces fatigue, and enhances cognitive performance at therapeutic doses ranging from 50-200 mg. The compound also stimulates cardiac muscle, relaxes smooth muscle, and produces diuretic effects through multiple pharmacological pathways.
Therapeutic vs. Toxic Doses
The therapeutic dose ranges from 50-200 mg for the desired stimulant effects. Typical medications contain 100-300 mg caffeine. Toxic doses in adults begin above 400 mg in a single dose, which may cause adverse effects, including anxiety and palpitations. Doses exceeding 1000 mg may cause serious toxicity with cardiac arrhythmias and seizure risk. The estimated lethal dose is 10-14 grams (150-200 mg/kg) for an average adult, with lower thresholds in children and sensitive individuals.
Acute Toxicity Symptoms
Low doses (50-200 mg) produce increased alertness, reduced fatigue, and mild stimulation. Moderate doses (200-400 mg) cause nervousness, restlessness, insomnia, tremors, and rapid heartbeat. High doses (above 400 mg) trigger severe anxiety, palpitations, nausea, vomiting, and cardiac arrhythmias. Toxic doses (above 1000 mg) can cause seizures, delirium, severe cardiac effects, respiratory failure, and potentially fatal outcomes.
Populations at Risk
Children are particularly sensitive to caffeine, with toxic effects occurring at lower doses than in adults. Pregnant women face reproductive and developmental effects risks. Individuals with cardiac conditions, anxiety disorders, or caffeine sensitivity experience adverse effects at lower doses. Concurrent use with other stimulants or medications increases the risk of adverse reactions and toxicity.
Technical Documents & References
Accurate documentation supports safe handling, quality control, and regulatory compliance for caffeine in various applications.
Safety Data Sheets (SDS) provide detailed safety information, physical properties, toxicological data, handling procedures, and emergency response information for caffeine anhydrous and specific formulations.
USP-NF Monographs specify pharmaceutical quality standards, purity requirements, analytical methods, and testing procedures for USP-grade caffeine used in drug manufacturing.
FDA Regulations detail permitted uses, maximum levels in foods and beverages, labeling requirements, and approved pharmaceutical applications across different product categories.
Certificate of Analysis (COA) verifies purity (typically 98-99% or higher), absence of contaminants, melting point, water content, and compliance with applicable standards (USP/NF pharmaceutical grade, food grade, or technical grade specifications) for each batch.
Final Note
Caffeine serves essential roles in pharmaceutical manufacturing, beverage production, dietary supplements, and research applications. Its stimulant properties make it valuable across numerous industries, yet these same properties create toxicity risks when handling concentrated forms.
Success with caffeine safety requires understanding the dramatic difference between therapeutic doses measured in milligrams and toxic doses measured in grams. A small amount of pure powder can contain dangerous quantities that look deceptively harmless. Maintaining dust control, preventing accidental ingestion through strict hygiene practices, and understanding dose-dependent toxicity symptoms form the foundation of safe caffeine handling.
Lab Alley supplies high-quality caffeine in pharmaceutical and technical grades meeting rigorous purity standards for diverse applications. Whether you're formulating medications, producing beverages, or conducting research, following the safety protocols in this guide helps you work confidently with this potent central nervous system stimulant while protecting workers from toxicity risks.
Explore our Resource Library for more information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need CaffeineYou Can Trust?
If you are looking for high-quality caffeine for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's caffeine is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.






