Shelf Life & Expiration of Caffeine: Storage and Stability Guide
Caffeine is a widely used alkaloid compound essential for pharmaceutical formulations, food and beverage production, dietary supplements, and cosmetic applications. Understanding how long it remains potent helps you maintain product quality and ensure consistent performance in critical applications. Caffeine is highly stable as a chemical compound, but proper storage practices prevent moisture absorption, clumping, and loss of potency that compromise effectiveness.
Caffeine stability depends on form, environmental conditions, and container selection. Anhydrous caffeine powder demonstrates exceptional longevity, while exposure to moisture, light, and heat accelerates degradation. This guide provides practical strategies for maximizing caffeine shelf life and identifying when replacement becomes necessary.


In this article, we’ll review:
- How Long Does Caffeine Last?
- Signs That Caffeine Has Expired
- What to Do If Caffeine Is Expired
How Long Does Caffeine Last?
Caffeine longevity is influenced by form, purity, packaging quality, and storage practices. Understanding these variables helps you make informed decisions about inventory management and replacement timing.
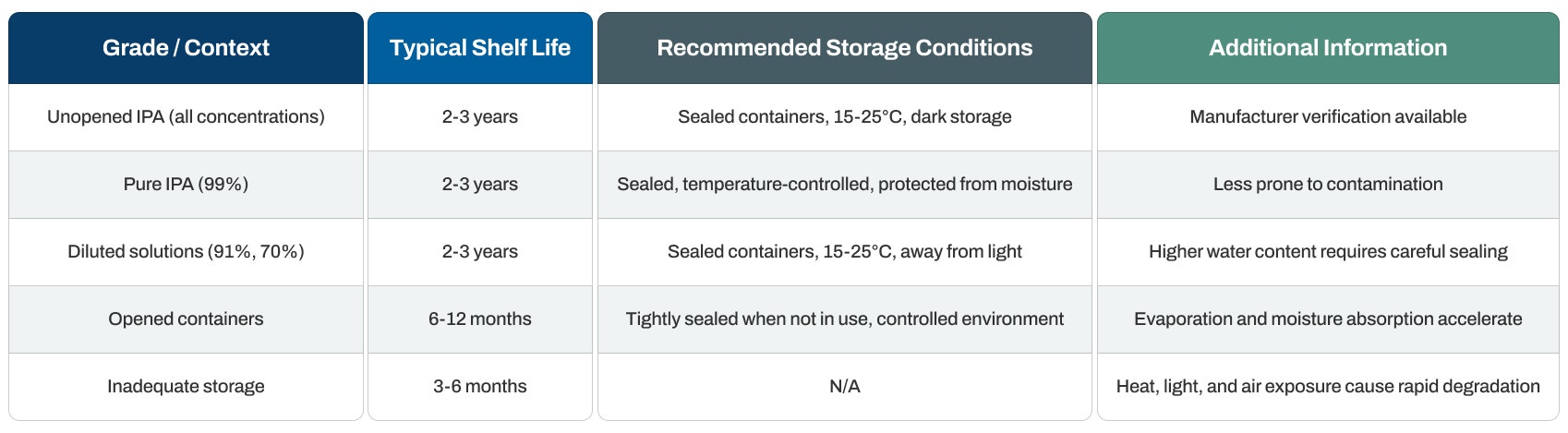
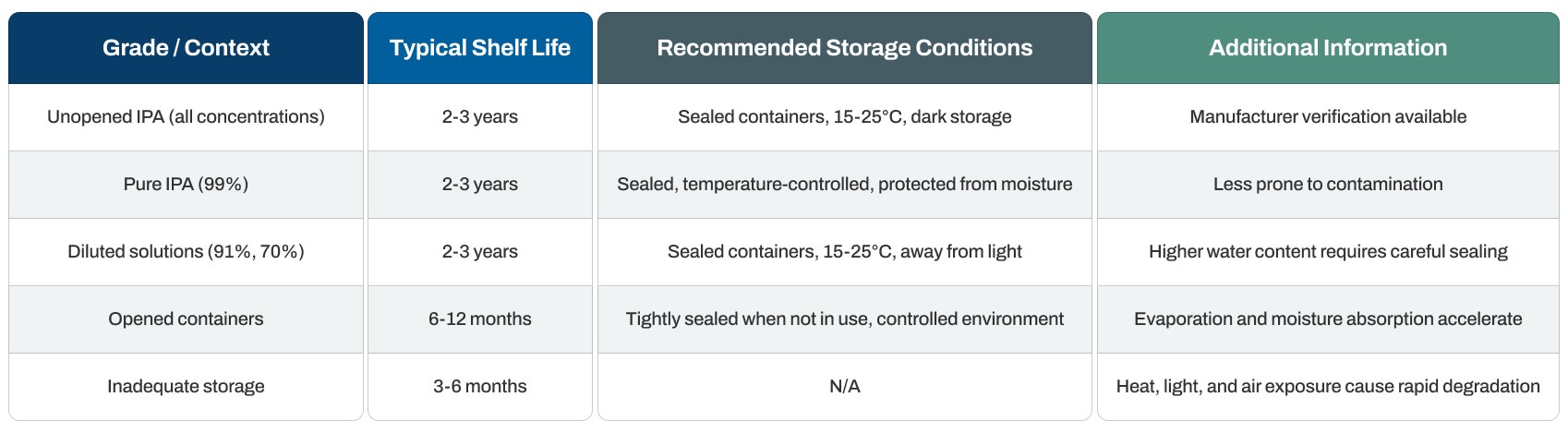
Shelf Life Duration
Unopened containers: Typically last 2 to 5 years from the manufacture date when stored correctly. This broad range reflects differences in form and storage conditions.
Pure anhydrous caffeine powder: Demonstrates particularly strong stability and can last up to 5 years or longer. The absence of water molecules in anhydrous caffeine ensures extended storage periods without risk of moisture-related degradation.
Pharmaceutical-grade caffeine: Generally follows the upper end of the shelf life range when sealed in moisture-proof containers, supporting long-term inventory for regulated applications.
Caffeine citrate formulations: May show shorter usable periods compared to anhydrous forms due to their hydrated nature and additional ingredients.
Food-grade caffeine: Shows comparable shelf life to pharmaceutical grades in sealed containers, though opened packages experience faster degradation from repeated air and moisture exposure.
Chemical Stability
Caffeine exhibits outstanding stability under typical conditions, though certain factors influence quality retention:
Molecular stability: Described as a stable, persistent molecule that doesn't degrade readily under proper storage conditions. This inherent robustness supports extended warehousing when handled correctly.
Degradation resistance: The chemical structure resists breakdown under normal circumstances, allowing reliable long-term storage for commercial applications.
Moisture sensitivity: Prolonged exposure to moisture, heat, and light can cause gradual degradation and loss of potency. While caffeine tolerates brief exposure, sustained contact with humidity triggers quality decline.
Water absorption: Hydrated forms or exposure to humid conditions can compromise stability. Anhydrous caffeine significantly outperforms hydrated versions in storage longevity.
Oxidation potential: Though less susceptible than many organic compounds, extended exposure to air combined with light and heat may trigger slow oxidative processes.
Purity & Packaging
Container selection and purity levels significantly affect how well caffeine maintains its quality:
Purity advantages: Higher purity pharmaceutical-grade caffeine in sealed, moisture-proof containers retains quality longer than food-grade or opened containers. Pharmaceutical specifications ensure a consistent composition that supports an extended shelf life.
Form differences: Anhydrous caffeine (pure caffeine powder without water molecules) demonstrates significantly greater stability than hydrated forms or caffeine citrate formulations. This distinction makes form selection crucial for long-term storage planning.
Container materials: Compatible materials such as HDPE, glass, and food-grade plastic provide adequate protection during storage. These materials resist chemical interaction and prevent contamination.
Moisture barriers: Containers must provide excellent moisture barrier properties to prevent water absorption. This characteristic becomes the most critical packaging consideration for caffeine storage.
Seal integrity: Tight sealing prevents both moisture ingress and air exposure. Exposure to air and moisture allows water absorption and contamination that accelerate degradation.
Original packaging: Manufacturer containers offer optimal protection until first use because they're engineered specifically for caffeine storage and transport.
Storage Conditions
Creating appropriate storage environments is fundamental to preserving caffeine quality:
Temperature guidelines: Maintain room temperature at 15-25°C (59-77°F) in a cool, dry location. This range prevents heat-induced degradation while avoiding unnecessary climate control expenses.
Humidity control: Store in low-humidity environments (below 60% relative humidity). Humidity proves particularly detrimental as it can cause caking, clumping, and potentially promote degradation. This represents the most critical environmental factor for caffeine storage.
Light shielding: Guard against direct sunlight and UV radiation. Store in cool, dark areas or opaque containers to prevent photodegradation that can reduce potency.
Heat avoidance: Excessive heat accelerates degradation processes. Position storage areas away from heat-generating equipment, windows, and direct sunlight.
Odor isolation: Keep away from strong odors, as caffeine can absorb odors from the environment. This characteristic requires separation from aromatic substances that could contaminate the product.
Contamination prevention: Keep away from strong bases, which can decompose caffeine. Isolate caffeine from incompatible substances that might trigger chemical reactions.
Quick Reference: Shelf Life and Storage
Signs That Caffeine Has Expired
Observable changes in appearance and performance indicate when caffeine has degraded beyond acceptable limits. Catching these signs early prevents using compromised material in critical applications.
Color Change: Yellowing or discoloration in normally white or off-white caffeine powder may indicate oxidation or degradation. Fresh caffeine should maintain its characteristic white or slightly off-white appearance.
Excessive Clumping or Caking: Clumping, caking, hardening, or changes in crystal structure suggest moisture absorption or degradation. While minor clumping may occur with handling, severe caking indicates significant moisture contamination.
Texture Alterations: Changes from free-flowing powder to compacted or sticky material point to water absorption affecting physical properties.
Gas Formation or Pressure Build-up: Extremely rare, but can indicate contamination or chemical decomposition in sealed containers. Swollen packages or pressure release upon opening require immediate investigation.
Reduced Effectiveness: Pharmaceutical formulations, beverages, or supplements may reveal reduced stimulant effects, altered dissolution rates, or inconsistent performance. Loss of expected physiological effects signals potency decline.
Loss of Bitter Taste: Caffeine possesses a characteristic bitter taste. Loss of this distinctive flavor may indicate degradation or contamination affecting chemical composition.
Altered Dissolution Behavior: Changes in how quickly caffeine dissolves in water or other
What to Do If Caffeine Is Expired
Discovering expired caffeine requires a systematic approach to safeguard product quality, maintain safety, and preserve performance.
Check Manufacturer Guidelines
Examine packaging labels and certificates of analysis for expiration or retest dates. Most packaging includes this information to guide proper usage, timing and quality assurance.
Pharmaceutical-grade caffeine should not be used past expiration for critical applications. Regulatory compliance in pharmaceutical settings demands strict observance of manufacturer-specified limits to ensure patient safety and product efficacy.
Food-grade and supplement applications may allow evaluation for continued use in less critical applications, though testing should confirm acceptable potency and purity before proceeding.
Follow Proper Disposal Procedures
Dispose of expired caffeine according to local regulations for chemical or pharmaceutical waste. While caffeine has relatively low environmental toxicity and is readily biodegradable, follow proper disposal protocols for commercial quantities to ensure regulatory compliance.
Small amounts from consumer products might go through regular waste channels where local codes permit, but verify requirements beforehand. Significant quantities from manufacturing or laboratory settings need handling according to chemical waste protocols.
Maintain disposal records according to good manufacturing practices or pharmaceutical standards. Documentation supports regulatory audits and quality system oversight.
Replace with Fresh Stock
Expired or degraded caffeine can be ineffective for pharmaceutical, food, or supplement applications. Source replacement caffeine from established suppliers who provide comprehensive documentation, including certificates of analysis showing recent manufacture dates.
Use fresh caffeine with a valid certification to ensure proper potency and consistent performance. Confirm that purity specifications, form (anhydrous vs. hydrated), and grade align with your application requirements, whether for pharmaceutical formulations, beverage production, dietary supplements, or cosmetic products.
Inspect incoming shipments for packaging integrity, proper sealing, and clear labeling. Verify that containers show no signs of moisture damage, clumping, or contamination before accepting delivery.
Proper Storage Extends Shelf Life
Implementing robust storage practices from the outset maximizes lifespan and preserves quality:
Container selection: Keep caffeine in tightly sealed containers made of compatible materials such as HDPE, glass, or food-grade plastic. Ensure containers provide excellent moisture barrier properties to prevent water absorption, the primary threat to caffeine stability.
Temperature management: Store at room temperature 15-25°C (59-77°F) in a cool, dry location. Avoid excessive heat, which can accelerate degradation and potentially trigger slow oxidative processes.
Light and heat shielding: Protect from direct sunlight and UV light exposure. Store in cool, dark areas or use opaque containers to prevent photodegradation that reduces potency over time.
Humidity control: Keep containers tightly sealed to prevent moisture absorption. Store in low-humidity environments (below 60% relative humidity). Moisture exposure leads to clumping, caking, and potential degradation, making humidity management the most critical storage consideration.
Contamination prevention: Use clean, dry utensils when handling to avoid introducing moisture or foreign substances. Dedicated scoops or dispensing equipment prevent cross-contamination.
Odor isolation: Keep away from strong odors, as caffeine can absorb odors from the environment. Store separately from aromatic substances, cleaning chemicals, or other materials with distinctive smells.
Chemical compatibility: Prevent cross-contamination with incompatible substances such as strong bases, which can decompose caffeine. Maintain separation from reactive chemicals in storage areas.
Proper labeling: Clearly label containers with contents, date received or opened, and expiration date to ensure proper rotation and timely use of stock. This practice supports first-in, first-out inventory management.
Inventory rotation: Use older stock first through organized inventory tracking. Monitor dates carefully to ensure material gets used while still within its optimal shelf life.
Condition monitoring: Regular inspection of storage environments catches problems before product quality deteriorates. Promptly address temperature fluctuations, humidity increases, or seal integrity concerns to maintain optimal conditions.
Form selection: For long-term storage needs, choose anhydrous caffeine powder over hydrated forms or caffeine citrate formulations. The absence of water molecules in anhydrous caffeine provides superior stability.
Conclusion
Caffeine typically lasts 2 to 5 years from manufacture when stored correctly, with pure anhydrous caffeine powder demonstrating exceptional stability up to 5 years or longer. Primary concerns include moisture absorption causing clumping and caking, gradual potency loss from prolonged exposure to humidity and heat, and potential odor absorption from the environment.
Appropriate storage in sealed, moisture-proof containers protected from heat, light, and humidity preserves quality and maximizes the lifespan of this stable alkaloid compound. Following manufacturer recommendations and implementing proper storage protocols ensures dependable performance across pharmaceutical, food and beverage, supplement, and cosmetic applications.
Note: Fresh caffeine stored under optimal conditions provides consistent potency and performance throughout its lifespan when handled according to manufacturer specifications and quality standards for your specific application.
For more detailed guidance, explore our Resource Library for comprehensive storage and handling information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Caffeine You Can Trust?
If you are looking for high-quality caffeine for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's caffeine is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.






