Citric Acid Safety & Hazards
Citric acid is an organic acid that is generally recognized as safe (GRAS) for food applications but can irritate skin, eyes, and the respiratory system in concentrated forms. While widely used in foods, beverages, pharmaceuticals, and cleaning products, this compound requires proper handling when working with concentrated solutions or powder forms.
Citric acid is available in countless everyday products, from soft drinks to household cleaners. Its GRAS status and use in dietary applications might suggest minimal risk, yet industrial-grade material and concentrated solutions present notable hazards. Direct contact with the eyes can cause serious irritation, and inhalation of dust may irritate the respiratory passages.
Citric acid is mildly acidic, so it may irritate with prolonged exposure; that’s why proper protective equipment and handling procedures remain important. While not classified as a carcinogen and considered relatively safe compared to other acids, understanding its hazard profile ensures safe use across laboratory, industrial, and commercial settings. This comprehensive guide covers everything necessary for working safely with citric acid.


In this article, we’ll explore:
- Chemical Hazards Overview
- Exposure Risks & Health Effects
- First Aid Measures
- Handling & Storage Precautions
- Spill & Leak Procedures
- Fire & Explosion Hazards
- Regulatory & Compliance Information
- Technical Documents & References
Chemical Hazards Overview
Citric acid presents a low to moderate hazard profile, significantly safer than many industrial acids. Its mild acidity and stable nature under normal conditions make it suitable for food applications, yet concentrated forms require attention to safety protocols.
Hazard Classification
Under the Globally Harmonized System (GHS), citric acid displays the exclamation mark pictogram (⚠️ GHS07), indicating its classification for causing skin irritation and serious eye irritation. This symbol warns of potential irritation effects and the need for basic precautions during handling.
The NFPA hazard rating system assigns citric acid specific risk levels: Health hazard rates as 1 (slight hazard), Flammability receives a 1 (slight fire hazard), and Reactivity is rated 0 (stable). These ratings reflect the compound's relatively benign nature compared to stronger acids and corrosive materials.
Citric acid is stable under normal conditions but mildly irritating in concentrated forms, requiring basic safety measures to prevent exposure-related issues.
Primary Risks
The main concerns from citric acid exposure come through several pathways, each presenting manageable risks with proper precautions:
-
Inhalation of dust: May cause respiratory tract irritation if inhaled as dust particles or mist from concentrated solutions. Workers handling powder forms face the highest exposure risk.
-
Skin contact: Concentrated solutions or prolonged contact with powder may cause mild skin irritation, redness, and dryness. The effects are generally less severe than with stronger acids.
-
Eye exposure: Can cause serious eye irritation with symptoms including redness, tearing, and discomfort. Direct contact with powder or concentrated solutions requires prompt attention.
-
Accidental ingestion: Generally recognized as safe in food applications; however, large amounts may cause gastrointestinal upset, including nausea, vomiting, and abdominal pain.
-
Physical hazards: Dust may create slip hazards on floors and work surfaces, particularly in areas where powder is frequently handled.
Exposure Risks & Health Effects
Understanding how citric acid affects the human body helps recognize situations requiring intervention. The severity of symptoms depends on concentration, duration of exposure, and individual sensitivity.
Inhalation
Citric acid may cause mild respiratory tract irritation from dust particles or concentrated solutions. Prolonged inhalation of dust may cause coughing, sneezing, and throat irritation. Workers in facilities producing or packaging citric acid face higher exposure risks from airborne particles.
Citric acid is generally well-tolerated at typical use concentrations found in food production and light industrial applications. Respiratory effects are typically mild and resolve quickly once exposure ceases. However, individuals with pre-existing respiratory conditions like asthma may experience more pronounced symptoms.
Skin Contact
Concentrated solutions of citric acid or prolonged contact with powder may cause mild skin irritation, redness, and dryness. The acidic nature of citric acid can disrupt the skin's natural barrier function, particularly with repeated exposure over time.
Repeated exposure may lead to skin sensitization in some individuals, though this occurs less frequently than with stronger acids. Workers who handle citric acid regularly should monitor for signs of developing sensitivity, including persistent redness or itching after contact. Dilute solutions are generally well-tolerated and pose minimal risk during normal handling.
Eye Contact
Citric acid causes eye irritation with symptoms including redness, tearing, and discomfort. Direct contact with powder or concentrated solutions may cause more severe irritation than dilute solutions. The crystalline nature of citric acid powder can cause mechanical irritation in addition to chemical effects.
Eye contact requires immediate attention to prevent more serious complications. While citric acid generally does not cause permanent damage with prompt treatment, delayed intervention can extend recovery time and increase discomfort.
Ingestion
Citric acid causes eye irritation with symptoms including redness, tearing, and discomfort. Direct contact with powder or concentrated solutions may cause more severe irritation than dilute solutions. The crystalline nature of citric acid powder can cause mechanical irritation in addition to chemical effects.
Eye contact requires immediate attention to prevent more serious complications. While citric acid generally does not cause permanent damage with prompt treatment, delayed intervention can extend recovery time and increase discomfort.
Chronic Effects
Citric acid is not classified as carcinogenic by regulatory agencies. Extensive toxicological studies and decades of widespread use support citric acid's safety profile for long-term exposure at typical use levels.
Citric acid is generally safe for long-term exposure at normal working concentrations. GRAS status indicates safety for regular consumption in foods, reflecting comprehensive safety evaluations. Workers who handle citric acid over extended periods can do so safely with basic protective measures.
Note: Most commercial citric acid is manufactured using Aspergillus niger fermentation. Some recent research has suggested potential sensitivity reactions in certain individuals, though this remains under investigation and appears to affect only a small subset of the population.
First Aid Measures
Quick and appropriate first aid response minimizes the effects of citric acid exposure. The specific treatment depends on the exposure route, but immediate action reduces symptom severity and promotes faster recovery.
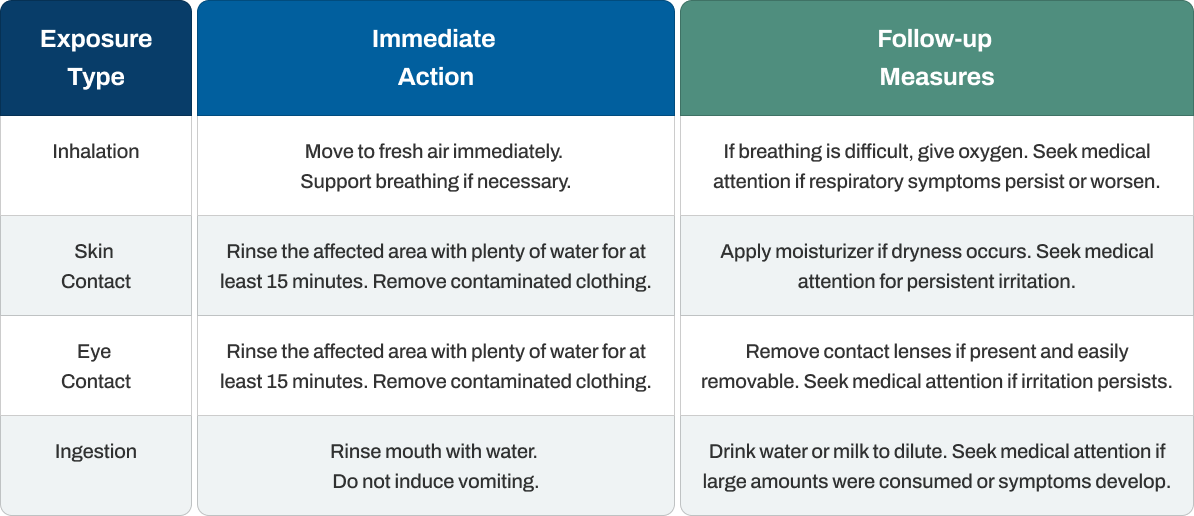

Critical considerations for first aid include using clean water for all rinsing procedures and providing medical personnel with specific information about the concentration and form of citric acid involved in the exposure. Time is particularly important with eye exposure, as delayed treatment can extend recovery time and increase discomfort.
Handling & Storage Precautions
Safe handling of citric acid requires basic precautions that address exposure prevention and product preservation. Proper procedures protect workers and maintain product quality over time.
Safe Handling Procedures
Use nitrile or latex gloves when handling large quantities of citric acid powder or concentrated solutions. While brief contact with dilute solutions poses minimal risk, gloves prevent prolonged exposure that can cause skin irritation.
Wear safety goggles when working with powders to prevent eye contact. The crystalline nature of citric acid powder makes eye protection particularly important during transfer operations, mixing, or any activity that might generate airborne particles.
Use dust masks when handling powder forms to minimize inhalation exposure. An N95 respirator or equivalent provides adequate protection for most citric acid handling operations. Ensure adequate ventilation in work areas to prevent dust accumulation and reduce airborne concentrations. Avoid generating airborne dust through careful handling techniques.
Storage Conditions
Store citric acid in a cool, dry, well-ventilated area away from incompatible materials. Keep containers tightly closed to prevent moisture absorption. Citric acid is hygroscopic and will absorb moisture from the air, leading to caking and potential quality issues.
Protect from excessive heat and humidity, which can cause caking of powder forms and degradation of product quality. Store at room temperature (15-25°C) in original containers whenever possible. Original packaging is designed for citric acid storage and includes appropriate labeling and safety information.
Incompatible Substances
Strong oxidizing agents and strong bases react with citric acid and should be stored separately. Metals such as aluminum, iron, and zinc, in the presence of moisture, can react with citric acid. The compound's chelating properties mean it actively binds with metal ions, potentially causing corrosion of metal containers or equipment.
Avoid contact with carbonates and bicarbonates, which can cause gas evolution (carbon dioxide) when mixed with citric acid. Incompatible with nitrates and chlorates, which may form explosive mixtures under certain conditions.
Spill & Leak Procedures
Citric acid spills require prompt attention to prevent exposure and minimize waste. The straightforward cleanup process reflects the compound's relatively low hazard profile compared to stronger acids.
Emergency Response Steps
Ensure adequate ventilation in the spill area to control dust formation from powder spills. Wear appropriate protective equipment, including a dust mask, gloves, and safety goggles. Avoid creating airborne dust during cleanup operations.
Containment Methods
Sweep up powder spills carefully to minimize dust generation. Use a dustpan and brush, or a vacuum equipped with appropriate filtration, to collect spilled material.
Absorb liquid spills with inert absorbent material such as vermiculite, sand, or commercial spill control products. Neutralize with sodium bicarbonate if necessary, particularly for large spills or concentrated solutions. Flush the area with water after cleanup to remove residual citric acid and prevent slip hazards.
Proper Disposal
Generally, biodegradable and can be disposed of according to local regulations. Citric acid breaks down readily in the environment through microbial metabolism. It can be neutralized before disposal to reduce the acidity of waste streams. Suitable for composting in small quantities. Avoid direct release to waterways in large concentrations.
Fire & Explosion Hazards
While citric acid presents minimal fire risk under normal conditions, understanding its behavior in fire situations ensures appropriate emergency response.
Flammability
Citric acid is a combustible solid that may contribute to fire intensity when involved in fires. Does not readily ignite under normal conditions, requiring significant heat input to reach ignition temperature. Powder may create explosive dust clouds under certain conditions when suspended in air at sufficient concentrations in the presence of an ignition source.
Extinguishing Media
Water spray, dry chemical, carbon dioxide, or foam are all suitable for fires involving citric acid. Water is effective for cooling and dilution. Avoid high-pressure water that may disperse powder and create additional hazards.
Firefighting Precautions
Use respiratory protection and full protective gear when fighting fires involving citric acid. Avoid inhalation of combustion products, which may include carbon monoxide and carbon dioxide. Citric acid may decompose to produce these gases when heated to decomposition temperatures.
Regulatory & Compliance Information
Citric acid is subject to various regulatory requirements that reflect its widespread use in food, pharmaceutical, and industrial applications.
OSHA Regulations
Citric acid requires standard chemical handling precautions and appropriate PPE use under OSHA guidelines. Generally considered a low-hazard classification compared to many industrial chemicals. Follows standard industrial hygiene practices, including adequate ventilation, dust control, and the use of personal protective equipment.
EPA & Environmental Impact
Low environmental hazard characterizes citric acid's environmental profile. Readily biodegradable and environmentally acceptable, citric acid breaks down rapidly through microbial action in soil and water. Minimal aquatic toxicity at typical use concentrations. Not bioaccumulative.
DOT Transportation Regulations
Generally not classified as hazardous material for transportation under Department of Transportation regulations. Ships as a non-hazardous commodity, requiring standard commercial shipping documentation without special hazardous materials handling requirements.
FDA/USP/ACS Compliance
GRAS status for food applications (E330 in the EU) confirms citric acid's safety for use in foods and beverages. USP grade available for pharmaceutical use meets United States Pharmacopeia standards for purity and quality. FCC grade for food applications ensures safety and purity standards. Meets international food additive specifications.
Note: Commercial citric acid is predominantly manufactured through Aspergillus niger fermentation rather than extracted from citrus fruits.
Technical Documents & References
Working with citric acid safely and effectively requires access to current technical documentation that provides detailed information about the specific product being used.
Safety Data Sheets (SDS)
Safety Data Sheets available from suppliers provide detailed safety, handling, and first aid measures specific to concentration and grade. Current SDS documents should be readily accessible to all workers handling citric acid.
GRAS Determination Documents
GRAS determination documents and toxicological studies support safety for food and cosmetic applications. These documents summarize decades of research on citric acid safety and provide the scientific basis for regulatory approvals.
Certificates of Analysis (COA)
Certificate of Analysis verifies product purity, moisture content, heavy metals, and compliance with food, pharmaceutical, or technical standards. COA documents are particularly important for pharmaceutical and food-grade applications where impurity levels must meet strict specifications.
Laboratories and facilities using citric acid should maintain organized files of technical documents for each product batch. Emergency contact information for poison control centers and environmental agencies should be readily available for quick expert assistance in case of incidents.
Final Note
Citric acid is a low-hazard chemical that requires basic handling precautions and proper safety procedures. Its GRAS status and widespread use in consumer products reflect its favorable safety profile, yet concentrated forms still demand respect and appropriate protective measures.
Success in citric acid safety comes from consistent application of basic procedures, adequate training, and maintaining current technical documentation. By following established protocols and using appropriate protective equipment when needed, you can safely work with this versatile organic acid across food, pharmaceutical, industrial, and consumer applications.
Explore our Resource Library for more information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Citric Acid You Can Trust?
If you are looking for high-quality citric acid for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's citric acid is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.










