How to Safely Dispose of Caffeine: Environmental and Safety Guidelines
Responsible disposal of caffeine serves a vital function in environmental stewardship, workplace safety, and meeting regulatory standards. This widely utilized compound appears in laboratories, pharmaceutical manufacturing, food and beverage production, research applications, and dietary supplement manufacturing, making proper disposal knowledge essential for professionals across diverse industries.
Caffeine disposal presents unique challenges based on waste characterization requirements, concentration levels, and pharmaceutical product regulations. Understanding appropriate disposal techniques protects workers from exposure to concentrated forms while ensuring compliance with federal and state regulatory frameworks.
This guide examines caffeine disposal fundamentals, addressing waste characterization protocols, regulatory obligations, approved disposal routes, and handling procedures appropriate for different product types and operational contexts.


In this article, we’ll review:
1. Why Proper Disposal Matters
2. How to Dispose of Caffeine
3. Special Considerations for Caffeine
Why Proper Disposal Matters
Caffeine disposal carries important implications that extend beyond routine waste handling. Understanding these consequences clarifies why regulatory agencies and facilities have established specific protocols for managing this compound.
Environmental Protection
Environmental considerations from caffeine occur when disposal practices fail to account for the compound's behavior in natural systems:
-
While caffeine is biodegradable and does not accumulate in aquatic organisms, it can contribute to water pollution when disposed of improperly. Caffeine has been detected in wastewater, surface waters, and groundwater due to the disposal of caffeinated beverages, pharmaceutical products, and industrial waste.
-
Improper disposal allows caffeine to reach aquatic ecosystems and drinking water sources. Although caffeine degrades relatively quickly in the environment with an atmospheric half-life of approximately 20 hours, concentrated releases can impact water quality before degradation occurs.
-
Biological treatment systems in wastewater facilities readily break down caffeine, yet concentrated solutions can overwhelm treatment capacity and pass through to receiving waters.
-
Caffeine in wastewater serves as an indicator of human waste contamination, making source reduction and proper disposal important for environmental monitoring and water quality protection.
-
Appropriate disposal prevents these environmental impacts and supports responsible chemical management that protects ecosystem health and drinking water quality.
Health & Safety
Human health and safety concerns make caffeine disposal a workplace priority, particularly in facilities handling concentrated forms:
-
Caffeine is moderately toxic with an oral LD50 in rats of approximately 192 to 367 mg/kg. Exposure to concentrated caffeine powder or solutions can cause acute toxicity, including central nervous system stimulation, cardiac arrhythmias, seizures, and in extreme cases, death.
-
Workers must avoid accidental ingestion of concentrated caffeine, particularly in pharmaceutical and laboratory settings where pure powder or concentrated solutions are handled. Even small amounts of pure caffeine can cause serious health effects.
-
Dust from caffeine powder can cause respiratory irritation when inhaled. Workers processing bulk caffeine powder face inhalation hazards that require dust control measures and respiratory protection.
-
Eye contact with caffeine powder or concentrated solutions causes irritation requiring immediate flushing. Direct contact requires prompt decontamination to prevent absorption.
-
Workers handling caffeine for disposal face hazards from accidental ingestion, inhalation of dust, and eye or skin contact. Establishing proper disposal procedures with suitable protective equipment is essential for preventing exposure incidents.
Legal Compliance
The regulatory structure governing caffeine disposal creates enforceable responsibilities based on waste characterization:
-
While caffeine itself is not listed as a hazardous waste under EPA regulations, waste generators must properly characterize caffeine-containing waste according to RCRA regulations (40 CFR 262.11) to determine if it exhibits hazardous characteristics, including ignitability, corrosivity, reactivity, or toxicity.
-
Pure caffeine powder or solutions typically do not meet RCRA characteristic waste criteria but must still be properly managed according to institutional protocols and local regulations.
-
Pharmaceutical caffeine products from healthcare facilities may require management under EPA Subpart P (40 CFR 266 Subpart P) for hazardous waste pharmaceuticals, which prohibits sewer disposal and mandates specific handling procedures.
-
Healthcare facilities subject to EPA Subpart P must segregate hazardous waste pharmaceuticals and follow specific labeling, storage, and disposal requirements that differ from general chemical waste protocols.
-
Industrial and laboratory facilities generating chemical waste are subject to federal, state, and local waste disposal regulations requiring compliance to avoid violations, fines, and legal liability.
-
Documentation requirements vary based on waste classification, generator status, and pharmaceutical product involvement, making proper waste characterization the foundation of legal compliance.
How to Dispose of Caffeine
Effective caffeine disposal begins with proper waste characterization and matching waste to appropriate disposal methods while meeting all regulatory requirements.
Characterize the Waste
Waste characterization forms the foundation of compliant caffeine disposal and determines applicable regulations.
Determine whether the caffeine waste is pure chemical powder, a pharmaceutical product, contaminated material, or mixed with other substances. This classification affects disposal options and regulatory requirements.
Review applicable RCRA regulations (40 CFR Part 261) to establish if waste exhibits hazardous characteristics, including ignitability, corrosivity, reactivity, or toxicity. Pure caffeine typically does not meet these criteria, but contaminated waste or mixtures may trigger hazardous waste classification.
Pharmaceutical caffeine products from healthcare facilities require evaluation under EPA Subpart P (40 CFR 266 Subpart P). These regulations govern hazardous waste pharmaceuticals differently from general chemical waste, with specific prohibitions and handling requirements.
Document waste composition, including caffeine concentration, product formulation, contaminants present, and origin. This documentation supports proper classification decisions and provides records for regulatory compliance verification.
Consider concentration when characterizing waste. Pure caffeine powder exceeding 98% purity requires more stringent handling than dilute solutions or formulated products. Always factor concentration into disposal method selection.
Check Applicable Regulations
Regulatory compliance verification establishes the groundwork for legally sound disposal operations.
Review federal EPA regulations governing chemical waste disposal, pharmaceutical waste management under Subpart P if applicable, and RCRA waste characterization requirements. Understand how these frameworks apply to your specific caffeine waste type.
Examine state environmental agency requirements, which may exceed federal standards. Some states impose additional restrictions on pharmaceutical waste disposal or chemical waste management that affect caffeine handling.
Research local waste disposal ordinances and wastewater discharge permits. Municipal regulations often contain specific prohibitions or authorization requirements for chemical waste disposal.
Verify if your facility is registered as a hazardous waste generator and determine generator status. Even facilities handling non-hazardous caffeine waste may need registration if other hazardous waste streams exist.
Check whether pharmaceutical waste regulations apply to caffeine-containing medications or supplements. Healthcare facilities face different requirements than research laboratories or manufacturing operations.
Consult institutional or company environmental health and safety protocols for chemical waste management. Organizations often maintain internal procedures that exceed regulatory minimums to ensure consistent waste handling.
Review the Safety Data Sheet (SDS) information for specific disposal recommendations based on product formulation and concentration. SDS provides manufacturer guidance that complements regulatory requirements.
Never Pour Down Drains
Drain disposal of caffeine is generally prohibited without proper authorization:
-
Caffeine should not be disposed of down sinks, drains, or toilets without explicit wastewater treatment authorization from the receiving facility.
-
Even though caffeine is biodegradable, concentrated solutions can overwhelm wastewater treatment systems. Biological treatment processes effectively remove caffeine at typical concentrations, but bulk disposal exceeds treatment capacity.
-
Healthcare facilities are prohibited under EPA Subpart P from disposing of pharmaceutical caffeine products down drains. This absolute prohibition applies to all pharmaceutical formulations regardless of concentration.
-
Bulk disposal to sewers violates Clean Water Act provisions and local wastewater discharge permits. Municipal authorities prohibit unauthorized chemical discharges that could impact treatment operations or receiving water quality.
-
Caffeine in wastewater serves as an indicator of human waste contamination, making source reduction important for environmental monitoring programs. Facilities should minimize caffeine contributions through proper waste segregation and disposal.
Use Designated Waste Disposal Methods
Appropriate disposal routes align with waste characteristics, product type, and regulatory classification.
Small Quantities (Laboratories and Research Facilities): Collect pure caffeine powder and solutions in clearly labeled chemical waste containers. Use containers compatible with the waste form, such as glass or plastic bottles with secure closures.
Store in designated chemical waste accumulation areas away from food and beverages. Maintain separation from laboratory operations to prevent accidental contamination or exposure.
Label containers with chemical name ("Caffeine"), concentration, hazard information, and accumulation start date. Include the responsible party names and contact information for institutional tracking.
Arrange pickup through institutional environmental health and safety programs or licensed chemical waste contractors. Academic and research institutions typically provide dedicated collection services with regular pickup schedules.
Pharmaceutical Quantities: Manage pharmaceutical caffeine products, including tablets, capsules, and injectable formulations, through pharmaceutical waste programs that comply with EPA Subpart P requirements.
Segregate hazardous waste pharmaceuticals according to regulatory criteria. Separate caffeine products that meet hazardous waste definitions from non-hazardous pharmaceutical waste streams.
Use reverse distributors for unused pharmaceutical products when applicable. Reverse distribution provides a legal pathway for returning unopened or partially used medications to manufacturers or authorized handlers.
Maintain proper documentation and waste manifests. Pharmaceutical waste requires detailed recordkeeping, including product names, National Drug Codes (NDC), quantities, and disposal methods.
Healthcare facilities must follow the EPA Subpart P prohibition on sewer disposal for pharmaceutical caffeine products. This applies to all concentrations and formulations managed within healthcare settings.
Industrial Quantities: Use bulk chemical waste collection services for manufacturing residues and process waste. Licensed waste contractors can handle drum quantities or bulk pickups from production facilities.
Consider recycling or recovery options for high-purity caffeine waste. Some waste contractors or specialty chemical companies may accept clean caffeine for reprocessing, particularly pharmaceutical or food-grade material.
Follow DOT transportation regulations for chemical waste shipment. While caffeine is not a DOT-regulated hazardous material, proper packaging and labeling support safe transport.
Maintain detailed waste characterization records and disposal certificates. Industrial operations require documentation demonstrating proper waste classification and disposal method selection.
Laboratory and Institutional Waste: Collect through institutional chemical waste programs with proper labeling showing chemical name, concentration, hazards, and accumulation start date.
Follow segregation protocols to prevent incompatible chemical mixing. Store caffeine waste separately from strong oxidizers, strong acids, and strong bases to prevent reactions.
Maintain chemical waste logs and disposal records for at least three years. Documentation should include waste generation dates, quantities, compositions, responsible parties, and disposal methods.
Adhere to institutional environmental health and safety protocols, which often exceed regulatory minimums to provide consistent standards across diverse chemicals and operations.
Approved Disposal Methods
Several disposal methods provide compliant pathways for caffeine waste based on characterization and applicable regulations.
Incineration: Pure caffeine and caffeine-containing waste can be dissolved or mixed with a combustible solvent and burned in a chemical incinerator equipped with an afterburner and scrubber.
High-temperature incineration ensures complete combustion and destruction of organic compounds. This method is preferred for pharmaceutical-grade caffeine waste and contaminated materials that cannot enter other disposal streams.
Licensed incineration facilities maintain appropriate air permits and pollution control equipment to manage combustion byproducts. Verify facility permits before arranging incineration disposal.
Licensed Waste Facility: Non-hazardous caffeine waste can be disposed of through licensed solid waste disposal facilities following state and local regulations.
Facilities must be permitted to accept chemical waste and maintain proper documentation. Verify acceptance criteria before shipping waste to ensure caffeine meets facility requirements.
This option works well for dilute caffeine solutions, formulated products, and materials that do not meet hazardous waste criteria but require segregation from general refuse.
Wastewater Treatment (With Authorization): Dilute aqueous caffeine solutions may be discharged to sanitary sewers only with prior written authorization from the receiving wastewater treatment plant.
Discharge must comply with local discharge permits, including concentration limits, pH requirements, and flow restrictions. Authorization typically specifies maximum caffeine concentrations and daily discharge volumes.
Caffeine is effectively removed by biological treatment processes in wastewater treatment plants operating properly. Municipal facilities can handle dilute caffeine within their design parameters.
This option requires written approval and ongoing adherence to concentration limits. Facilities must maintain records demonstrating compliance with authorized discharge parameters.
Never Mix Chemicals
Chemical compatibility concerns influence safe disposal practices and prevent hazardous reactions.
Avoid mixing caffeine waste with strong oxidizing agents, including hydrogen peroxide, nitric acid, and potassium permanganate. These combinations can cause exothermic reactions or generate heat.
Keep caffeine waste separate from strong acids and strong bases. Although caffeine is relatively stable, mixing with concentrated acids or bases can cause decomposition or unwanted reactions.
Maintain separation from incompatible materials during storage and disposal. Use dedicated containers for caffeine waste and never combine with other chemical waste streams without verifying compatibility.
Chemical reactions from improper mixing can generate heat, produce hazardous decomposition products, or create disposal complications that increase costs and regulatory scrutiny.
Follow PPE Guidelines
Worker protection during caffeine disposal requires appropriate personal protective equipment:
-
Always wear nitrile or latex gloves when handling caffeine powder or concentrated solutions. Gloves prevent dermal contact and potential absorption through skin.
-
Use safety goggles or glasses to protect eyes from powder or solution splashes. Eye protection is essential during all handling operations, including waste transfers and container filling.
-
Don lab coats or protective clothing to prevent contamination of personal clothing and minimize skin exposure. Dedicated laboratory attire should remain in work areas.
-
Use dust masks or respirators when working with caffeine powder to avoid inhalation of airborne particles. Respiratory protection prevents inhalation exposure during weighing, transferring, or handling bulk powder.
-
Ensure adequate ventilation in work areas. Local exhaust ventilation or general room ventilation prevents dust accumulation and reduces airborne exposure.
-
Have eyewash stations and safety showers available when handling bulk quantities. Emergency decontamination equipment
Special Considerations for Caffeine
Caffeine disposal involves unique factors based on product type, concentration, and regulatory classification.
Pharmaceutical Products
Caffeine in pharmaceutical formulations, including tablets, capsules, and solutions, must be managed according to pharmaceutical waste regulations.
Healthcare facilities subject to EPA Subpart P must segregate hazardous waste pharmaceuticals and follow the prohibition on sewer disposal. This applies to all pharmaceutical caffeine products regardless of concentration or formulation.
Controlled substances containing caffeine as combination products require DEA compliance for disposal. Facilities handling controlled substance formulations must follow DEA regulations in addition to EPA requirements.
Use reverse distributors for unused pharmaceutical products when applicable. Reverse distribution provides a compliant pathway for managing excess inventory or recalled products.
Maintain detailed documentation, including product names, National Drug Codes, quantities, and disposal methods. Pharmaceutical waste requires more extensive recordkeeping than general chemical waste.
Laboratory and Research Settings
Universities and research institutions should manage pure caffeine through institutional environmental health and safety chemical waste programs.
Label caffeine waste containers with chemical name, concentration, accumulation date, and hazard information. Include the responsible researcher's names and laboratory locations for institutional tracking.
Store in designated chemical waste accumulation areas following institutional protocols. Separate from active laboratory operations and maintain secure access to prevent unauthorized handling.
Research facilities typically provide dedicated collection programs with regular pickup schedules. Utilize established institutional programs rather than arranging independent disposal.
Analytical standards and laboratory-grade caffeine should be consolidated in compatible waste containers. Small quantities can be combined, provided waste characterization remains accurate.
Food and Beverage Industry
Food-grade caffeine waste from manufacturing must be characterized and disposed of according to facility waste management plans.
Dilute aqueous waste streams may be pretreated through on-site wastewater systems with proper permits. Industrial pretreatment programs allow authorized discharge after treatment to meet municipal acceptance criteria.
Concentrated caffeine solutions require collection and disposal through licensed waste contractors. Bulk concentrations exceed wastewater treatment capacity and require segregated handling.
Manufacturing residues and process waste should be characterized to determine if recovery or recycling is feasible. High-purity waste may have value for reclamation operations.
Household and Consumer Quantities
Over-the-counter caffeine supplements and medications should be disposed of through household hazardous waste collection programs or pharmaceutical take-back events.
Do not flush down toilets or throw in regular trash. Proper disposal prevents environmental contamination and accidental exposure to children or pets.
Many pharmacies and law enforcement agencies offer medication disposal programs. These convenient collection points accept unused medications, including caffeine-containing products.
Consumer quantities are exempt from federal hazardous waste regulations under the household hazardous waste exclusion, yet still require proper disposal through designated collection programs.
Contaminated Materials
Lab equipment, filters, gloves, and other materials contaminated with concentrated caffeine should be managed as chemical waste.
Small quantities of contamination may allow disposal as non-hazardous waste after proper characterization. Document waste classification decisions to demonstrate compliance with characterization requirements.
Materials heavily contaminated with pure caffeine powder require segregated disposal. Treat as chemical waste requiring incineration or licensed facility disposal.
Cleaning and decontamination may allow some materials to enter general waste streams. Evaluate contamination levels and document decisions regarding waste classification.
Emergency Spills and Response
Caffeine spills require prompt response to minimize exposure and environmental release.
For caffeine powder spills, avoid creating dust. Use vacuum equipment with HEPA filtration or carefully sweep material into waste containers. Minimize air disturbance during cleanup.
For solution spills, absorb with inert absorbent materials and collect in waste containers. Vermiculite, absorbent pads, or commercial spill products work well for liquid cleanup.
Dispose of spill cleanup materials as caffeine-contaminated waste. Contaminated absorbents and cleaning materials require the same disposal pathway as the original caffeine waste.
Ensure adequate ventilation during cleanup operations. Open doors and windows or use mechanical ventilation to prevent dust accumulation.
Documentation and Recordkeeping Requirements
Maintaining comprehensive records demonstrates regulatory compliance and supports waste characterization decisions.
Keep waste manifests, characterization records, and disposal certificates for a minimum of three years. Some states require longer retention periods, so verify local requirements.
Document decisions regarding hazardous versus non-hazardous waste classification. Characterization records should include reasoning, data sources, and responsible party signatures.
Maintain Safety Data Sheets on file for all caffeine products handled. SDS provides essential information for waste classification, emergency response, and worker protection.
Retain disposal records demonstrating proper waste handling. Documentation should include disposal methods, contractor information, facility permits, and compliance certifications.
Track waste generation quantities to determine generator status and reporting obligations. Monthly tracking supports accurate regulatory classification and permits compliance.
Quick Reference: Disposal Methods and Safety Requirements
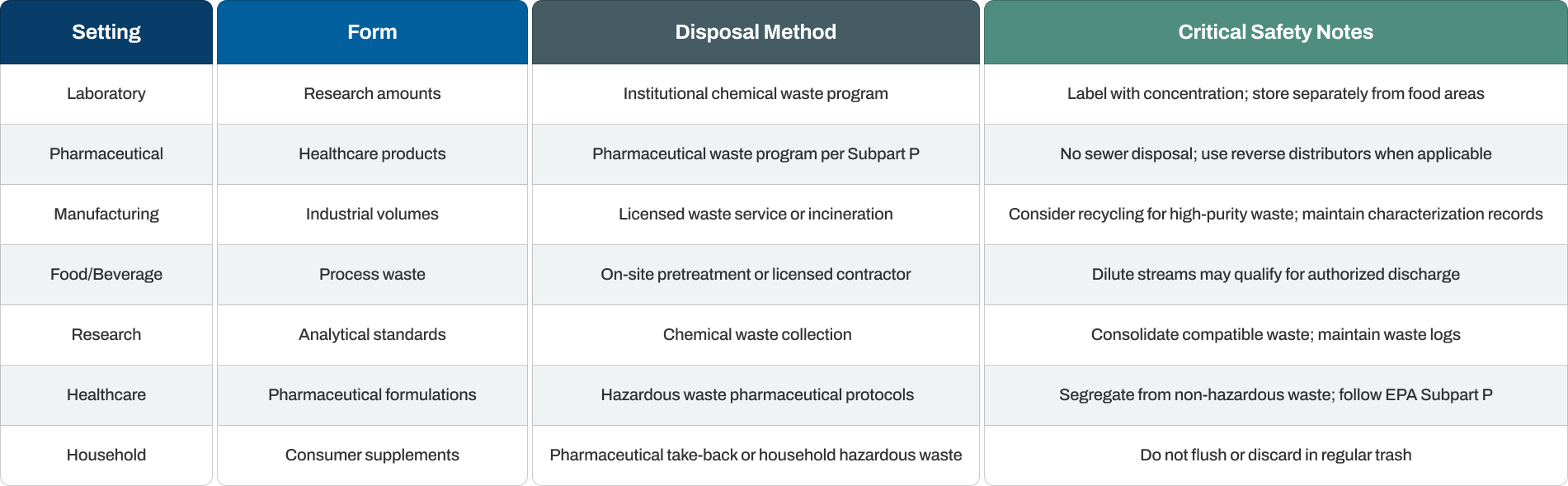
Final Note
Caffeine waste characterization determines applicable disposal requirements, with pharmaceutical products facing stricter regulations than pure chemical or food-grade materials. Understanding waste characterization protocols, EPA Subpart P requirements where applicable, and approved disposal methods promotes compliant, environmentally responsible disposal operations.
For safe, regulatory-compliant disposal of caffeine waste and expert guidance on characterization and pharmaceutical waste requirements, contact institutional environmental health and safety departments, licensed chemical waste disposal contractors, or pharmaceutical waste management services. Always consult current Safety Data Sheets (SDS) for specific handling and disposal information related to your particular caffeine product and formulation, and follow all applicable institutional, company, federal, state, and local regulations for chemical waste management.
When uncertain about disposal procedures or waste characterization, consult environmental health professionals or waste disposal specialists for guidance specific to your product type, concentration, volume, and jurisdiction.
For more detailed guidance, explore our Resource Library
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Caffeine You Can Trust?
If you are looking for high-quality caffeine for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's caffeine is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.






