Lactic Acid Shelf Life & Expiration: Storage and Stability Guide
Lactic acid is a versatile organic acid used across food manufacturing, cosmetics, pharmaceuticals, and industrial processes. Understanding its shelf life and proper storage requirements ensures you maintain product quality and effectiveness throughout its useful life. While relatively stable compared to many organic compounds, lactic acid requires attention to storage conditions to prevent degradation and contamination.
The stability of lactic acid depends heavily on concentration, storage environment, and packaging. Concentrated solutions show remarkable longevity under proper conditions, while dilute formulations face greater challenges from microbial contamination. This article provides comprehensive guidance on maximizing lactic acid shelf life and recognizing when replacement becomes necessary.


In this article, we’ll review:
- How Long Does Lactic Acid Last?
- Signs That Lactic Acid Has Expired
- What to Do If Lactic Acid Is Expired
How Long Does Lactic Acid Last?
Lactic acid shelf life depends on multiple factors, including grade, concentration, packaging, and storage environment. Understanding these variables helps you predict longevity and implement appropriate storage strategies.
Shelf Life Duration
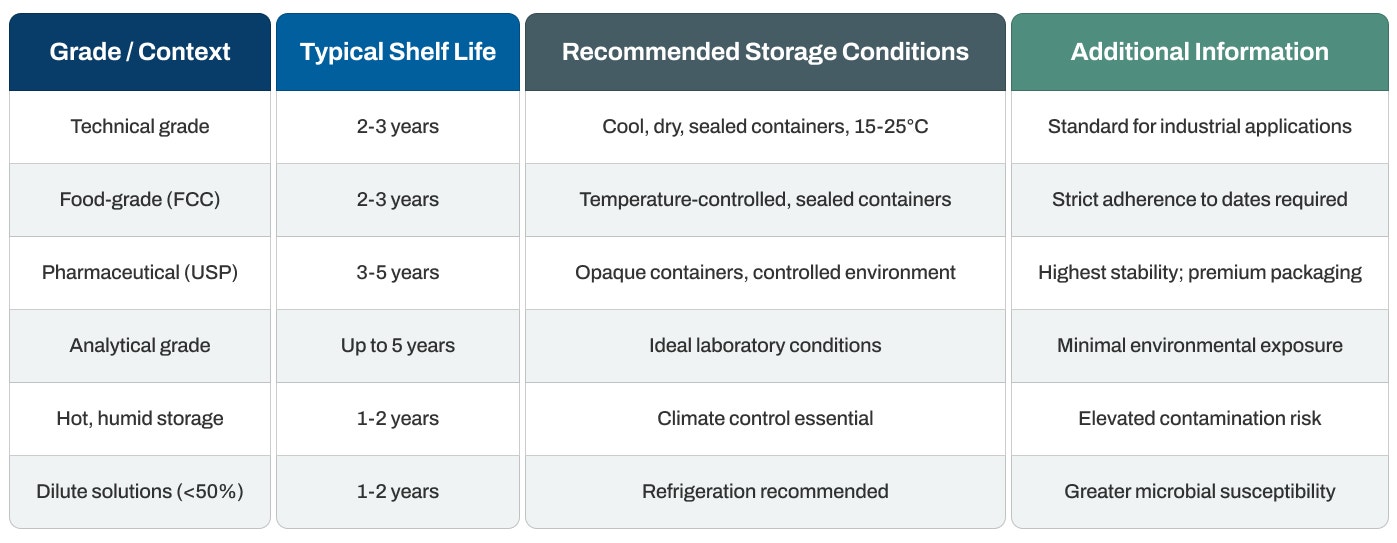
- Technical and food-grade solutions: Typically maintain effectiveness for 2-3 years when stored in optimal conditions with proper sealing and temperature control.
- Pharmaceutical grade (USP): Can maintain stability for 3-5 years when properly stored in sealed containers away from light and temperature extremes. The higher purity standards and careful packaging of pharmaceutical grades support extended storage.
- Climate impact: In hot, humid climates or poor storage conditions, the shelf life may be reduced to 1-2 years due to potential bacterial contamination or concentration changes from evaporation.
- High-purity analytical grades: Can maintain stability for up to 5 years when stored under ideal laboratory conditions with minimal exposure to environmental factors.
Chemical Stability
Lactic acid solutions demonstrate good stability under normal conditions, though several factors influence degradation rates:
- Baseline stability: The compound does not readily decompose under normal storage conditions, making it suitable for long-term inventory management when environmental controls are adequate.
- Concentration effects: Higher concentrations (80-90%) demonstrate superior stability compared to dilute solutions. The reduced water content and higher acidity of concentrated solutions inhibit microbial growth that can compromise product quality.
- Temperature sensitivity: Elevated temperatures above 30°C promote polymerization reactions, forming lactide or higher molecular weight compounds. These polymers alter solution properties and reduce effectiveness in applications requiring monomeric lactic acid.
- Light-induced degradation: Gradual degradation occurs with light exposure, particularly in clear containers. Photochemical reactions cause slight discoloration and can alter chemical composition over extended periods.
- Concentration drift: pH changes may occur over time due to water evaporation, which concentrates the solution. This affects applications requiring precise acidity levels and can impact formulation consistency.
Purity & Packaging
Packaging choices significantly influence how long lactic acid maintains its quality:
- Container materials: Glass or high-density polyethylene (HDPE) containers provide optimal protection for long-term storage. Metal containers risk corrosion from acidic solutions, potentially contaminating the product and compromising container integrity.
- Purity advantages: Higher purity grades in sealed, opaque containers retain quality longer than lower grades in permeable packaging. Pharmaceutical and analytical grades benefit from stringent purity specifications that minimize impurities, catalyzing degradation.
- Bulk storage systems: Large-scale operations typically employ stainless steel tanks with proper ventilation and temperature control. These systems accommodate industrial volumes while maintaining environmental conditions supporting product stability.
- Seal integrity: Proper sealing prevents evaporation and concentration changes that alter product specifications. Even small leaks allow moisture exchange and concentration drift that affects quality over time.
Storage Conditions
Environmental management forms the foundation of successful long-term lactic acid storage:
- Temperature range: Maintain storage areas at 15-25°C (59-77°F) for optimal stability. This range prevents thermal degradation while inhibiting microbial growth that could compromise dilute solutions.
- Light protection: Protect from direct sunlight and UV exposure through opaque containers or storage in dark areas. Photochemical degradation proceeds slowly but accumulates over extended storage periods.
- Temperature extremes: Avoid both excessive heat and freezing temperatures. Heat accelerates chemical reactions and supports microbial growth, while freezing may cause physical separation or container damage.
- Chemical segregation: Store away from strong bases, oxidizing agents, and materials that could introduce contamination. Incompatible materials can trigger reactions degrading product quality or creating safety hazards.
- Sealing protocol: Ensure containers remain tightly sealed to prevent evaporation and concentration changes. Regular inspection confirms seal integrity throughout the storage period.
Quick Reference: Shelf Life and Storage
A quick and appropriate first aid response can significantly reduce the severity of lactic acid exposure injuries. Immediate action remains the priority for all exposure routes.

Signs That Lactic Acid Has Expired
Visual and physical changes indicate when lactic acid has degraded beyond acceptable quality levels. Early recognition prevents the use of compromised material that could affect your processes or products.
- Color Change: Development of yellow, brown, or amber coloration in normally clear solutions signals degradation or contamination. Fresh lactic acid appears clear to very pale yellow; significant color changes indicate quality loss.
- Crystallization or Precipitation: Formation of crystals, sediment, or particulate matter that persists even after gentle warming demonstrates chemical instability or contamination affecting product usability.
- Concentration Changes: Significant deviation from the labeled concentration occurs through evaporation or water absorption, affecting pH and effectiveness. Applications requiring precise acidity suffer when concentration drifts outside specifications.
- Microbial Contamination: Visible mold growth, cloudiness, or unusual odors indicate bacterial or fungal contamination, particularly in dilute solutions. Biological contamination renders products unsuitable for any application, especially food and cosmetic uses.
- Viscosity Changes: Unusual thickening or polymerization, especially in concentrated solutions exposed to heat, signals molecular changes through polymerization reactions that alter product characteristics.


What to Do If Lactic Acid Is Expired
Discovering expired lactic acid requires a systematic response to protect product quality and ensure regulatory compliance across your applications.
Check Manufacturer Guidelines
Consult packaging labels and certificates of analysis for expiry or retest dates. Food-grade and pharmaceutical applications demand strict adherence to expiration dates for safety and regulatory compliance. Industrial applications may allow retesting by qualified personnel to determine continued suitability, though food and pharmaceutical uses prohibit this flexibility.
Follow Proper Disposal Procedures
Neutralize expired lactic acid with sodium bicarbonate or lime before disposal according to local regulations. The compound's biodegradability and low environmental toxicity simplify disposal compared to many industrial acids, though proper neutralization remains important.
Avoid discharging concentrated solutions to drains without neutralization, as temporary pH alterations can affect wastewater systems. Label disposal containers clearly and work in ventilated areas when handling expired material. Consult local waste management authorities for region-specific disposal requirements.
Replace with Fresh Stock
Source replacement lactic acid from reputable suppliers providing comprehensive documentation, including certificates of analysis with current dates. Verify grade and purity specifications match your application requirements, whether for food preservation, cosmetic formulations, pharmaceutical products, or industrial processes.
Inspect incoming shipments for packaging integrity, proper sealing, and clear storage recommendations. Confirm concentration specifications align with your process needs before accepting delivery.
Keep in Mind: Proper Storage Extends Shelf Life
Implement robust storage protocols from the beginning to maximize shelf life: Use appropriate containers constructed from glass or HDPE that resist acid corrosion. Original manufacturer packaging often provides optimal protection until first use.
Maintain storage temperatures between 15-25°C while protecting from light through opaque containers or dark storage areas. Ensure tight sealing to prevent evaporation and concentration drift. Rotate inventory systematically to use older stock first, preventing accumulation of aging material. Regular environmental monitoring identifies conditions requiring adjustment before product quality suffers.
Conclusion
Lactic acid generally lasts 3-5+ years for concentrated pharmaceutical-grade solutions with excellent inherent stability. Technical and food-grade solutions typically provide 2-3 years of reliable service. Proper storage in appropriate containers away from heat and light maximizes the useful life of this stable organic acid while ensuring consistent quality for your applications.
Note: Fresh lactic acid stored under optimal conditions delivers reliable performance throughout its shelf life when handled according to safety guidelines and manufacturer specifications for your particular application.
For more detailed guidance, explore our Resource Library.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Lactic Acid You Can Trust?
If you are looking for high-quality lactic acid for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's lactic acid is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.








