Non-Denatured Ethanol Overview: Properties, Applications, Safety & Sourcing Guide
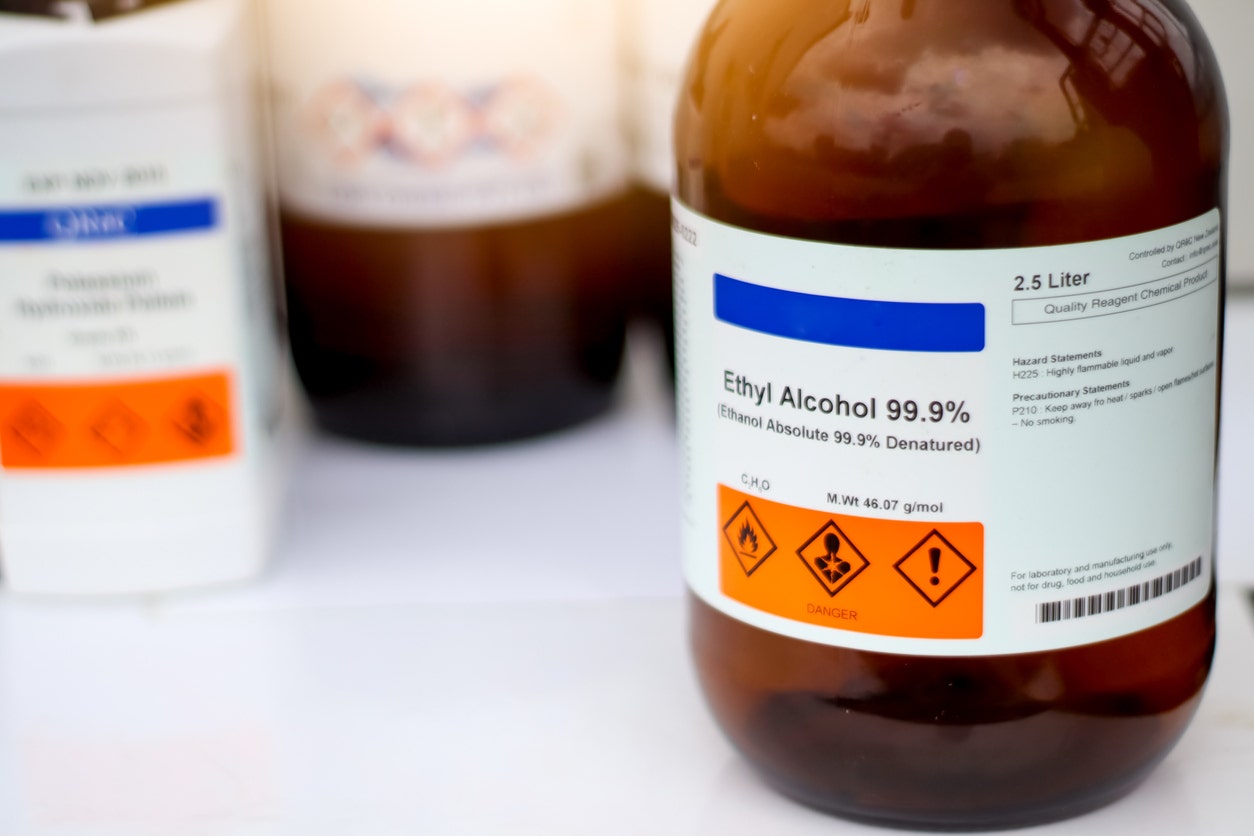
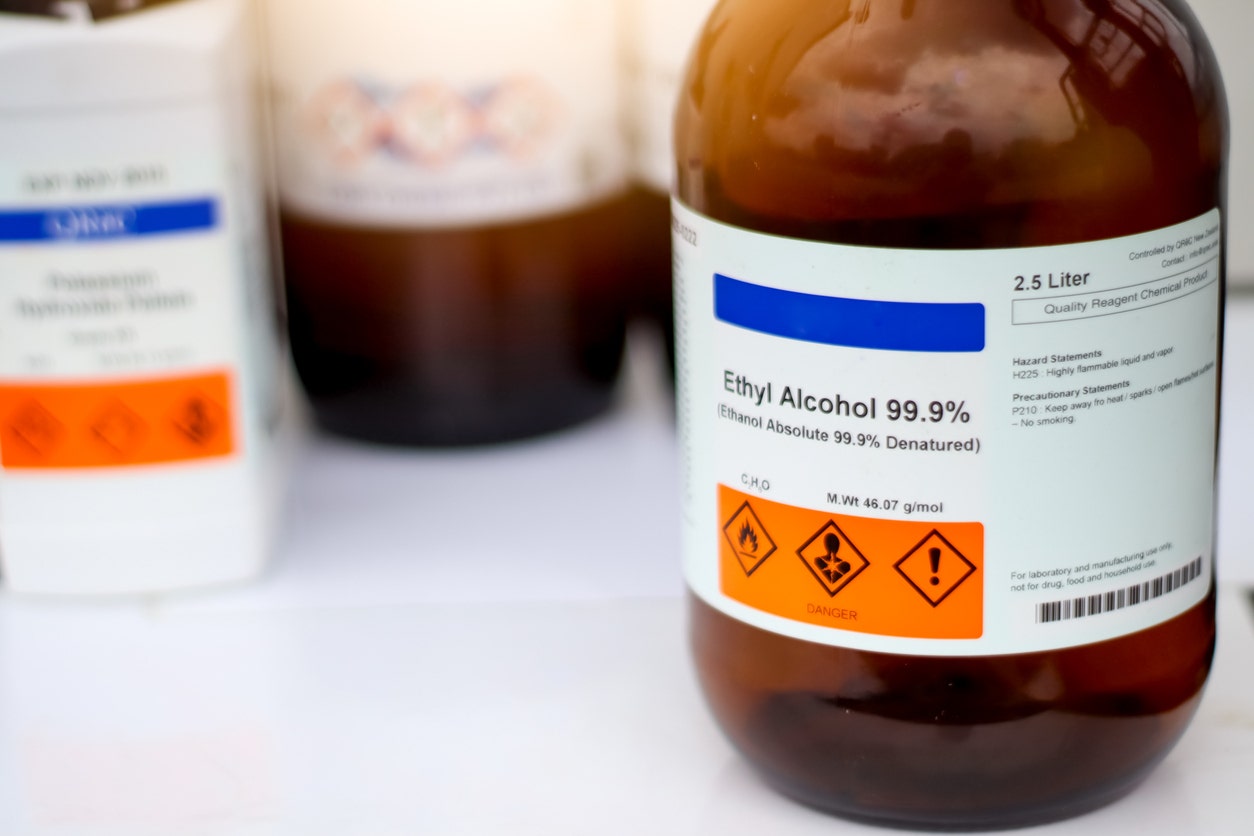
Non-denatured ethanol, also known as pure ethyl alcohol or undenatured ethanol, is a clear, colorless, volatile organic compound with the chemical formula C₂H₆O. Unlike its denatured counterpart, this version contains no toxic additives or denaturants, making it the only choice when purity and safety matter most.
This pure form of ethanol serves as the foundation for pharmaceutical manufacturing, where it dissolves active ingredients in medications. Craft distilleries build their operations around non-denatured ethanol, transforming it into premium spirits. Food processing facilities extract vanilla, herbs, and botanical flavors using this solvent. Research laboratories rely on it for DNA extraction, tissue processing, and analytical procedures.
The key difference between non-denatured and denatured ethanol comes down to what's been added. Denatured versions contain poisonous additives like methanol or isopropanol that make the liquid undrinkable. Non-denatured ethanol has nothing added, which means it can safely contact food, enter pharmaceutical formulations, and serve medical purposes. This purity carries high costs, though. Federal excise taxes and strict regulatory controls make non-denatured ethanol substantially more expensive and harder to obtain than denatured alternatives.
Understanding non-denatured ethanol's properties and regulations becomes essential whether you're formulating pharmaceuticals, producing food extracts, manufacturing beverages, or conducting research. This guide covers everything from chemical characteristics to purchasing requirements.
Properties of Non-Denatured Ethanol
The molecular structure of non-denatured ethanol creates characteristics that make it invaluable across industries while demanding careful handling and regulatory compliance.
Key Facts About Non-Denatured Ethanol
-
Compound Name: Non-denatured ethanol
-
Synonyms: Pure ethyl alcohol, Undenatured ethanol, Grain alcohol, Absolute alcohol, Cologne spirit, EtOH, Spirits of wine, Food-grade ethanol, Pharmaceutical ethanol, UN 1170
-
Chemical Formula: C₂H₆O (CH₃CH₂OH)
-
CAS Number: 64-17-5
-
Molar Mass: 46.07 g/mol
-
Appearance: Clear, colorless liquid with characteristic ethereal, vinous odor
Density Characteristics
At 20°C, non-denatured ethanol weighs approximately 0.789 g/cm³, making it lighter than water. This lower density affects how you measure volumes, size equipment, and respond to spills. The density difference also matters during emergency response since floating ethanol spreads across water surfaces rather than sinking.
Thermal Properties
Non-denatured ethanol boils at 78.37°C (173.1°F), well below water's boiling point. This explains why ethanol evaporates so quickly at room temperature. The melting point sits at -114.6°C (-174.3°F), keeping ethanol liquid even under extreme cold.
The flash point of 12.8°C (55°F) dominates all safety planning. Standard room temperature exceeds this threshold. Non-denatured ethanol constantly releases flammable vapors at typical working temperatures. A tiny spark, static electricity, or hot equipment surface can ignite these vapors instantly.
Ethanol and water create an azeotrope at 95.6% concentration. At this ratio, both compounds evaporate together at identical rates. Simple distillation cannot push concentrations higher. Reaching absolute ethanol above 99% requires specialized dehydration techniques using molecular sieves.
Non-denatured ethanol is a high-purity alcohol used in food, beverage, pharmaceutical, cosmetic, laboratory, and industrial applications. It supports extraction, sanitation, formulation, preservation, and chemical synthesis where no toxic additives are allowed. U.S. laboratories, manufacturers, and processors sourcing non-denatured ethanol by grade and proof level can contact Lab Alley at 512-668-9918 for product selection and ordering support.
Solubility and pH
Mix non-denatured ethanol with water in any proportion and they blend completely. This unlimited miscibility simplifies formulation work. Most organic solvents also dissolve readily in ethanol. Fats and oils resist dissolving, which becomes useful when extracting specific compounds while leaving lipids behind.
Pure non-denatured ethanol measures approximately pH 7.33, sitting near neutral. Unlike acids or bases, ethanol won't corrode metals through pH attack or irritate tissues through extreme acidity or alkalinity.
Reactivity and Stability Considerations
Strong oxidizers create violent reactions with non-denatured ethanol. Hydrogen peroxide, nitric acid, and similar compounds produce rapid reactions that generate enough heat to ignite ethanol instantly. Strong acids also create dangerous situations with violent boiling or ignition.
Mix ethanol vapors with air and explosive atmospheres form between 3.3% and 19% vapor concentration. This wide range demands constant vigilance about vapor accumulation in enclosed spaces.
Non-denatured ethanol pulls moisture from the air. Open a bottle of absolute ethanol in a humid environment and the concentration gradually drops as atmospheric water gets absorbed. High-purity grades need immediate resealing after use.
Ethanol fires produce nearly invisible pale blue flames barely visible in daylight. People have walked into ethanol fires without seeing flames until feeling intense heat.
Under normal storage conditions, stability remains excellent. Properly sealed non-denatured ethanol sits unchanged for years. The main concern becomes concentration drift through evaporation or moisture absorption if containers aren't perfectly sealed.
Non-denatured ethanol from Lab Alley is offered in food, pharmaceutical, cosmetic, technical, and high-purity analytical grades, with concentrations from 190 proof to 200 proof and packaging options ranging from small lab bottles to 55-gallon drums.
Applications & Benefits
Food & Beverage Applications
Spirits production starts with high-proof non-denatured ethanol diluted to the desired strengths. Vodka, gin, whiskey, rum, and other spirits rely on beverage-grade ethanol as their foundation. The beverage alcohol industry consumes enormous volumes while facing comprehensive TTB regulation.
Flavor extraction pulls aromatic compounds from vanilla beans, herbs, spices, and botanical materials. High-proof non-denatured ethanol dissolves molecules that water cannot extract. Commercial vanilla extract production depends entirely on food-grade ethanol. Hundreds of flavoring agents use ethanol as the carrier solvent.
Medical & Pharmaceutical Applications
Antiseptics for surgical preparation typically use 70% non-denatured ethanol, providing rapid kill against bacteria, viruses, and fungi. The 70% concentration proves more effective than higher concentrations because residual water helps denature microbial proteins.
Hand sanitizers in healthcare settings rely on 60% to 70% ethanol concentrations. Healthcare workers sanitize their hands dozens of times daily between patient contacts. Non-denatured formulations avoid skin irritation from toxic denaturants.
Pharmaceutical solvents dissolve active ingredients throughout drug manufacturing. Many oral liquid medications use ethanol as the vehicle carrying drugs that won't dissolve in water alone. Pharmaceutical processing uses ethanol during tablet granulation, controlled-release coating, and synthesis of active ingredients.
Laboratory & Research Uses
General-purpose solvent applications make non-denatured ethanol nearly ubiquitous in research laboratories. It cleans glassware without residue, dissolves samples for analysis, and participates in countless procedures.
DNA and RNA precipitation represent a fundamental molecular biology technique using cold ethanol. Adding ethanol to nucleic acid extracts causes DNA or RNA to precipitate as visible strands for purification.
Histology tissue processing moves biological samples through a graded ethanol series for dehydration. Pathology laboratories process thousands of tissue samples through an ethanol dehydration series. Chromatography mobile phases and equipment sterilization round out common applications.
Cosmetic & Personal Care
Premium perfumes dissolve fragrances and essential oils in non-denatured ethanol that evaporates cleanly after application. The absence of denaturants prevents unpleasant chemical odors from interfering with expensive fragrance compounds.
Personal care antiseptics include mouthwashes, toners, and aftershaves incorporating ethanol for antimicrobial properties and astringent action. Products that make skin contact benefit from using non-denatured grades that avoid irritation from toxic additives.
Industrial Applications
Pharmaceutical manufacturing represents the primary industrial use, where non-denatured ethanol serves as a high-value solvent throughout production. Chemical synthesis operations producing pharmaceutical intermediates sometimes require non-denatured grades when denaturants interfere with reactions.
Specialized cleaning applications requiring maximum purity specify non-denatured ethanol despite costs. When cleaning pharmaceutical equipment or preparing surfaces for sterile operations, avoiding denaturant contamination justifies premium pricing.


Fight Your Kidney Stone with This Natural Product Hack
Fight kidney stones. Discover how citric acid naturally prevents stone formation.


Is Lactic Acid Bad? A Look at the Health Science
Is lactic acid bad for you? Debunking myths and revealing its vital role in your body & beyond.


Hospitality Without Hitchhikers: Boric Acid’s Role in Bed Bug Prevention
Protect your hospitality business. Boric acid is key to preventing bed bug infestations.


Harnessing Silica Sulfuric Acid to Enhance Concrete Durability
Enhance concrete longevity. Silica sulfuric acid improves strength and chemical resistance.
Safety Information
Health Hazards & Precautions
Flammability dominates all safety considerations. The 12.8°C (55°F) flash point sits well below normal room temperature. Non-denatured ethanol constantly releases flammable vapors. Classification as a Class IB flammable liquid reflects serious fire risks requiring comprehensive controls.
Health effects from vapor inhalation include eye and respiratory tract irritation at moderate concentrations. Higher vapor levels cause headaches, dizziness, drowsiness, and central nervous system depression. Skin contact causes drying and irritation with repeated exposure. Eye contact causes stinging and potential corneal damage.
The nearly invisible flame presents exceptional danger. Pale blue flames remain barely visible in well-lit work areas. Vapor density exceeding air means ethanol vapors sink and accumulate in low areas like basements and floor drains.
Compared to denatured alternatives containing methanol or other toxic additives, non-denatured ethanol presents lower toxicity risks. The absence of poisonous denaturants reduces hazards from skin contact and accidental ingestion.
Personal Protection Requirements
Safety goggles or chemical safety glasses protect eyes from liquid splashes. Chemical-resistant nitrile gloves prevent skin contact and drying. Lab coats or chemical-resistant aprons shield clothing and skin from splashes. Closed-toe shoes protect feet from spills.
Respiratory protection becomes necessary when ventilation cannot control vapor levels below exposure limits. Organic vapor cartridges on respirators filter ethanol vapors.
Storage Guidelines
Cool, dry, well-ventilated storage areas minimize vapor generation. Approved flammable liquid storage cabinets provide critical fire protection. OSHA regulations limit storage to 60 gallons per cabinet with a maximum of three cabinets per fire area.
Seal containers tightly immediately after use. Ground and bond containers during transfers to prevent static electricity. Maintain physical separation from oxidizers and strong acids.
Emergency Procedures
Fire response demands alcohol-resistant foam extinguishers specifically designed for polar solvents. Standard protein foam breaks down on alcohol contact. CO₂ and dry chemical extinguishers also work for small fires.
Spill response starts with ignition source elimination. Shut off electrical equipment, remove hot objects, and clear the area. Absorbent materials soak up spilled ethanol for disposal as hazardous waste. Ventilation during cleanup prevents vapor accumulation reaching explosive concentrations.
Exposure first aid involves removing persons from vapor sources to fresh air. Flush affected skin with water for at least 15 minutes while removing contaminated clothing. Eye exposure requires continuous flushing with water for 15 minutes. Seek medical attention for persistent symptoms, significant ingestions, or eye injuries.
Regulatory Overview
OSHA Classification
Class IB flammable liquid designation triggers extensive workplace safety requirements, including approved storage cabinets, proper labeling, fire protection systems, and worker training. The permissible exposure limit sits at 1000 ppm time-weighted average for eight-hour work shifts.
DOT Classification
UN 1170 identifies ethanol for shipping purposes. Hazard Class 3 designates flammable liquids, requiring specific packaging meeting DOT specifications. Packing Group II reflects a moderate danger level. Proper shipping papers must accompany all shipments.
TTB Regulations
The Alcohol and Tobacco Tax and Trade Bureau oversees non-denatured ethanol under federal alcohol regulations. Federal excise tax applies at $13.50 per proof gallon for tax-paid ethanol. A gallon of pure ethanol generates $27.00 in taxes.
Industrial use permits allow qualified institutions to obtain non-denatured ethanol tax-free for approved purposes. Permit holders must maintain detailed records tracking receipt, use, and disposition. Beverage alcohol applications face comprehensive TTB licensing requirements.
Age restrictions limit non-denatured ethanol purchases to individuals 21 years and older.
FDA Regulations
GRAS status for food applications appears in 21 CFR 184.1293, confirming safety when used according to good manufacturing practices. Food-grade ethanol purchases require documentation confirming FCC certification.
Pharmaceutical applications face USP standards defining purity requirements. Current good manufacturing practices govern pharmaceutical production using ethanol.
Environmental Considerations
Biodegradability makes non-denatured ethanol environmentally favorable. Microorganisms readily metabolize ethanol, breaking it down to carbon dioxide and water. This rapid biodegradation means environmental persistence remains low. Renewable source availability from grain and sugarcane provides sustainability advantages over petroleum-derived solvents.


How to Safely Dispose of Sulfuric Acid
Sulfuric acid is one of the most common acidifying reagents and sulfur sources, essential across multiple industries. At the same time, it is also a toxic and highly hazardous chemical reagent, requiring safe handling and disposal by trained personnel.


How To Use Nitric Acid Safely
Nitric acid is a corrosive chemical and harmful to humans. Its contact with skin and eyes can cause serious burns. Affected skin will require flushing with soap and lukewarm water for at least 30 minutes.


Acetone Safety: Proper Storage and Disposal Methods
Acetone is a staple chemical solvent in most laboratories. It is also perhaps one of the most recognizable chemicals to the layperson, best known as the pungent solvent comprising nail polish remover.


How to Use Potassium Iodide Safely and Where to Buy it Online
Potassium Iodide Powder, also known as Potassium Salt of Hydriodic Acid, Iodic Acid Potassium Salt, Potassium Monoiodide, and Knollide, has the chemical formula KI. It appears as a white crystalline powder, odorless, and is soluble in water at ambient conditions.


Medicinal Oils Derived From Plants
You can order food-grade ethanol, extraction-grade ethanol, ACS-grade ethanol, FCC-grade ethanol, USP grade ethanol, HPLC-grade ethanol and kosher ethanol from Lab Alley. Perform ethanol (EtOH) extraction at ultra-low temperatures for the best yields.
Ethanol extraction is a cost-effective way to use ethanol as an industrial solvent to produce high-quality botanical extracts from large volumes of medicinal herbs or plants.
Non-Denatured Ethanol Grades and Pack Sizes
Available Grades & Purities
Multiple pharmaceutical-grade options include 200 proof absolute ethanol at 99.5% minimum, 190 proof at 95%, and various intermediate concentrations. Food-grade ethanol conforming to FCC specifications comes in similar ranges. Some suppliers offer dual USP/FCC certification. Beverage-grade ethanol at regulated proof levels serves spirits production. ACS and reagent grades provide analytical purity for research. Organically certified ethanol maintains organic integrity throughout production.
Pack Sizes & Bulk Availability
Small laboratory containers ranging from 500ml to 4 liters suit research facilities and low-volume users. Medium containers at 5 gallons or 20 liters serve regular users and small manufacturing operations. Bulk quantities, including 55-gallon drums, 270-gallon totes, and tank truck deliveries, accommodate high-volume industrial users and pharmaceutical manufacturers.
Common concentrations include 70% for disinfection applications, 95% for general use, and 99%+ for anhydrous applications.
Regulatory Requirements
Beverage alcohol applications require TTB permits for production, distribution, and sale. Industrial use permits provide tax-free access to qualified institutions for approved purposes. Tax-paid ethanol purchases avoid permit requirements but include federal excise taxes. Age verification confirms that the purchaser meets minimum age requirements of 21 years old.
Advantages Over Denatured Ethanol
The absence of toxic denaturants represents the fundamental advantage justifying higher costs. No methanol, isopropanol, or other poisonous additives means non-denatured ethanol can safely contact food, enter pharmaceutical formulations, and serve medical purposes. Denatured ethanol cannot substitute in these applications.
Higher purity levels become available with non-denatured grades. Pharmaceutical and analytical grades offer purity specifications impossible with denatured alternatives. Safety for human contact applications reduces hazards in healthcare settings, personal care products, and food processing. Renewable production from grain or sugarcane provides environmental advantages.









