Non-Denatured Ethanol Shelf Life & Expiration
Non-denatured ethanol is a highly stable organic solvent with GRAS status that maintains excellent longevity when stored properly. However, its effectiveness in food, pharmaceutical, and analytical applications depends on protecting it from evaporation, moisture absorption, and light exposure. Understanding how long non-denatured ethanol remains viable and compliant with regulatory standards helps ensure consistent quality and safety across all applications.
Does non-denatured ethanol expire? While exceptionally stable chemically, it can lose effectiveness through concentration changes, moisture contamination, and light-induced degradation. This article explains typical shelf life expectations, warning signs of expiration, and proven storage methods to maximize useful life while maintaining food-grade and pharmaceutical compliance.


In this article, we’ll review:
- How Long Does Non-Denatured Ethanol Last?
- Signs That Non-Denatured Ethanol Has Expired
- What to Do If Non-Denatured Ethanol Is Expired
How Long Does Non-Denatured Ethanol Last?
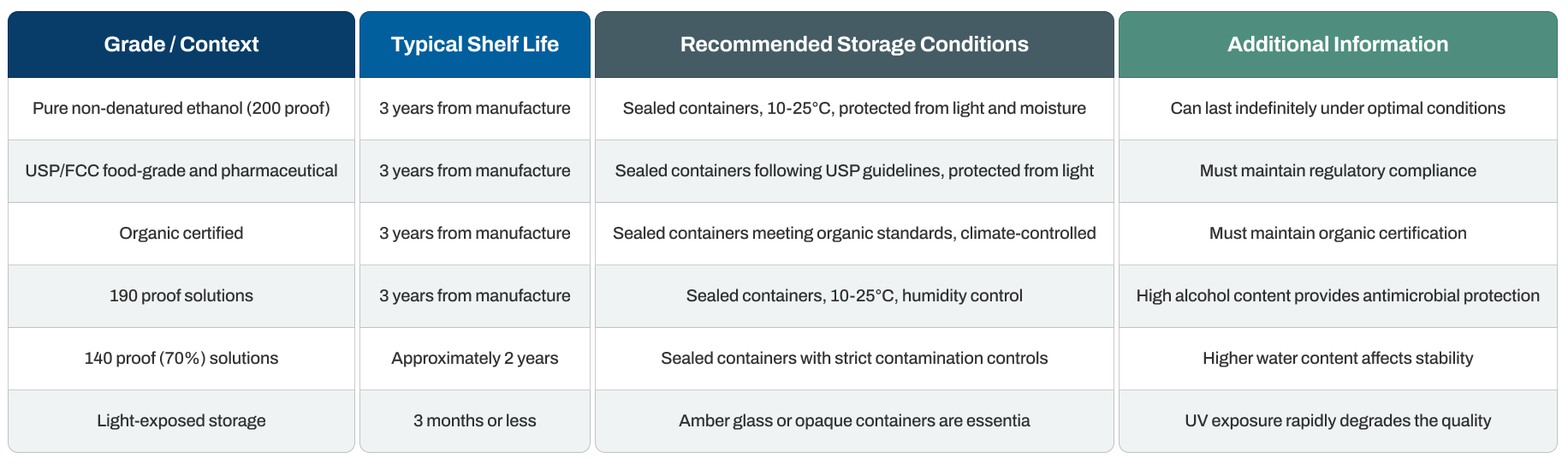
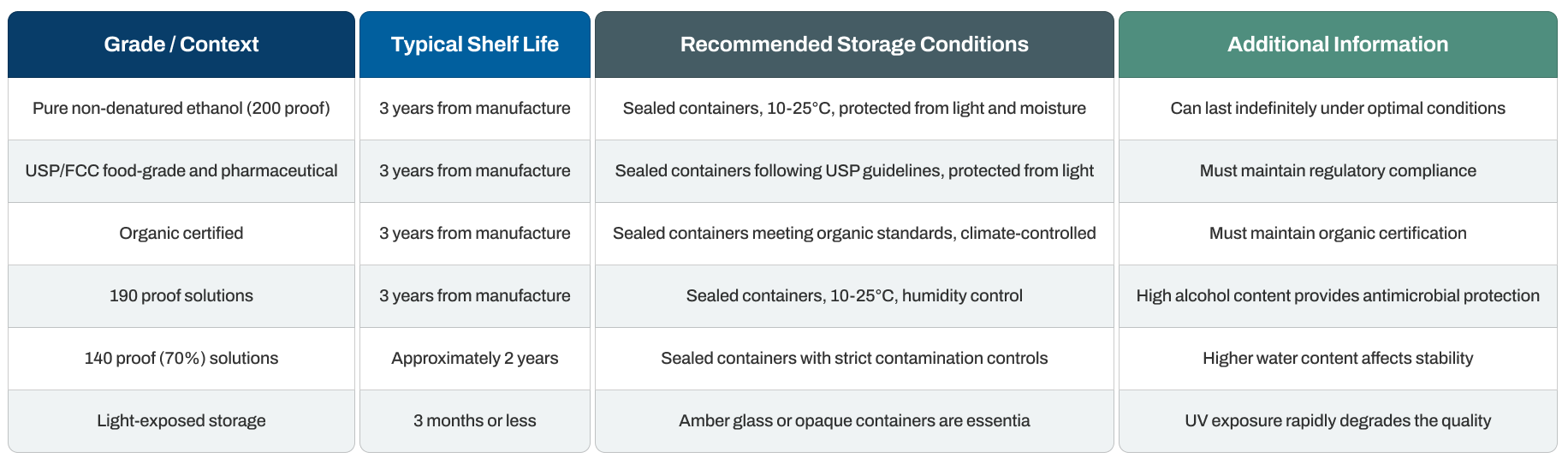
Non-denatured ethanol shelf life varies based on proof, packaging, and storage conditions, with pure grades typically maintaining quality and regulatory compliance for three years from manufacture when stored in optimal conditions. However, environmental factors like light exposure, temperature, and container integrity significantly impact this timeline.
Chemical Stability
The chemical stability of non-denatured ethanol makes it one of the most reliable solvents for long-term storage:
- Exceptional stability: Non-denatured ethanol is exceptionally stable and does not readily decompose under normal storage conditions, maintaining its purity for food and pharmaceutical applications. This stability stems from its simple molecular structure and lack of reactive functional groups.
- Natural antimicrobial properties: Pure non-denatured ethanol acts as a natural antimicrobial agent with GRAS status, preventing microbial growth and contamination in food-grade applications. This self-preserving quality contributes to its long shelf life.
- Minimal degradation risk: Can theoretically degrade to form acetaldehyde under extreme conditions with heat and light exposure, but this is unlikely during proper storage and does not affect food-grade status under normal conditions.
- Evaporation concerns: The primary concern is evaporation rather than chemical degradation, given its relatively low boiling point of 78.37°C (173°F). This can affect concentration and regulatory compliance, particularly in partially filled or imperfectly sealed containers.
- Light sensitivity: UV light exposure can cause pure non-denatured ethanol to fail within 3 months, depending on packaging, potentially affecting USP and food-grade specifications. This makes light protection critical for maintaining quality.
Purity & Packaging
The grade and packaging of non-denatured ethanol directly impact how long it remains effective and compliant with regulatory standards:
- High purity advantage: Higher purity grades (190-200 proof) in sealed, food-grade containers retain quality and regulatory compliance longer than diluted solutions. The lower water content reduces opportunities for microbial growth or chemical interactions.
- Food-grade materials required: Storage must use food-grade materials such as HDPE plastic, glass, and stainless steel to maintain food safety and USP compliance. These materials don't leach contaminants or react with the ethanol.
- Avoid reactive materials: Never use untreated aluminum or certain brass alloys that may corrode, leach contaminants, or compromise food-grade status. Material compatibility verification is essential before selecting storage containers.
- Original containers optimal: Original sealed containers provide optimal protection against evaporation, contamination and maintain traceability for regulatory compliance. Manufacturer packaging is specifically designed for long-term ethanol storage.
Storage Conditions
Environmental conditions represent the most significant factor influencing non-denatured ethanol longevity and regulatory compliance:
- Temperature control: Best kept in a cool, dry, well-ventilated area at 10-25°C (50-77°F) following USP controlled room temperature guidelines for pharmaceutical applications. Temperature extremes can accelerate evaporation and affect container integrity.
- Light and heat protection: Protect from direct sunlight, heat sources, and UV light exposure to maintain chemical stability and food-grade specifications. Use amber glass or opaque containers when possible for light-sensitive applications.
- Fire safety requirements: Store away from ignition sources, sparks, and electrical equipment due to high flammability, following food processing facility safety standards. Proper separation distances and ventilation are mandatory.
- Humidity control: Maintain relative humidity below 50% to prevent moisture absorption, as non-denatured ethanol is hygroscopic. This is particularly important for absolute ethanol (200 proof), which readily absorbs water from the air.
- Proper sealing is essential: Ensure containers are tightly sealed to prevent evaporation, concentration changes, and maintain regulatory compliance for food and pharmaceutical applications. Even small openings can lead to significant concentration drift over time.
Quick Reference: Shelf Life and Storage
A quick and appropriate first aid response can significantly reduce the severity of ethanol exposure injuries. Immediate action remains the priority for all exposure routes.
Signs That Non-Denatured Ethanol Has Expired
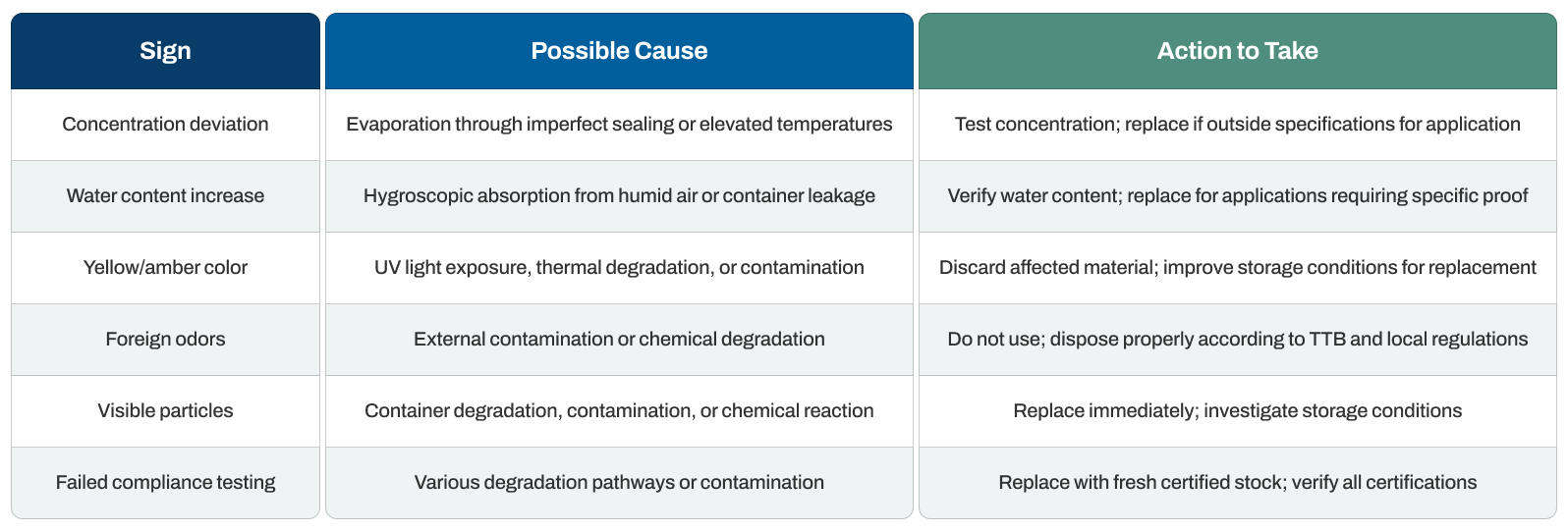
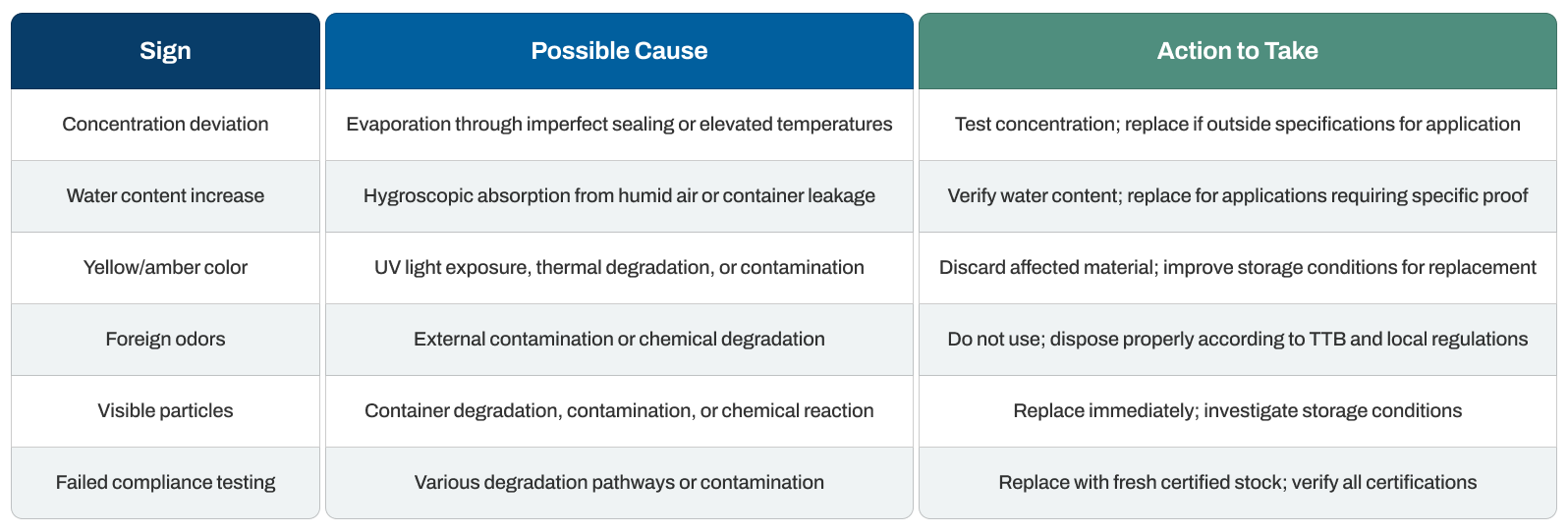
Non-denatured ethanol displays clear indicators as it deteriorates or loses compliance with regulatory standards. Recognizing these signs early prevents reduced effectiveness and ensures safety in food, pharmaceutical, and analytical applications.
- Concentration Changes: Significant deviation from labeled proof or percentage due to evaporation, affecting effectiveness and regulatory compliance in food, pharmaceutical, extraction, and analytical applications. Even small concentration changes can impact critical processes.
- Moisture Contamination: Evidence of water absorption leading to lower alcohol content, particularly concerning for food-grade applications where precise concentrations are required for safety and efficacy. Absolute ethanol is especially vulnerable to moisture pickup.
- Discoloration: Development of yellow or amber coloration in normally clear non-denatured ethanol, indicating degradation, contamination, or compromise of food-grade quality standards. Fresh non-denatured ethanol should be crystal clear.
- Unusual Odors: Off-smells or foreign odors that weren't present in fresh non-denatured ethanol, suggesting contamination or degradation that may compromise food safety or pharmaceutical purity. Pure ethanol has a characteristic, clean, sharp odor.
- Sediment or Particulates: Formation of visible particles, cloudiness, or foreign matter in the solution, indicating contamination that compromises food-grade and pharmaceutical standards. This suggests either container degradation or external contamination.
- Container Integrity Issues: Cracks, leaks, or degraded seals that compromise the ethanol's purity, concentration, and food-grade compliance, potentially leading to contamination. Container damage often indicates improper storage conditions.
- Regulatory Compliance Issues: Failure to meet USP, FCC, organic, kosher, or halal specifications upon retesting, affecting suitability for food, pharmaceutical, and regulated applications. Regular quality verification may reveal compliance failures even without visible changes.
To quickly assess your non-denatured ethanol's condition, refer to the table below:
What to Do If Non-Denatured Ethanol Is Expired
When non-denatured ethanol shows signs of expiration or fails quality standards, follow established procedures to ensure safety, regulatory compliance, and maintain the effectiveness of your applications.
Check Manufacturer Guidelines
Always start by consulting supplier recommendations and official documentation. Follow expiry or retest dates provided on packaging or certificate of analysis. Food-grade, pharmaceutical USP, and organic certified applications must strictly adhere to expiration dates and regulatory compliance standards.
For industrial applications with less stringent requirements, some suppliers include retesting protocols that can help determine whether borderline material remains suitable for specific uses. However, this should only be done by qualified personnel with appropriate testing capabilities and should never apply to food or pharmaceutical applications.
Follow Regulatory Compliance Assessment
Verify continued compliance with USP, FCC, organic, kosher, and halal certifications through appropriate testing protocols. Expired non-denatured ethanol may no longer meet food safety or pharmaceutical purity requirements even if it appears unchanged.
Testing should verify concentration accuracy, water content, absence of contaminants, and any other parameters specified in the relevant standards. Documentation of test results is essential for regulatory compliance and traceability.
Use Approved Disposal Procedures
Disposal must comply with multiple regulatory frameworks, given non-denatured ethanol's unique status. Dispose of according to local hazardous waste regulations and TTB requirements for non-denatured ethanol. This material must be handled as both a flammable liquid and a regulated beverage alcohol.
Never pour down drains or dispose of in regular trash, as this violates both environmental and TTB regulations. The flammable nature and regulatory status require special handling throughout the disposal process.
Contact authorized waste disposal services familiar with beverage alcohol regulations. Standard hazardous waste contractors may not be equipped to handle the TTB compliance aspects of non-denatured ethanol disposal.
Collect expired material in clearly labeled containers designed for flammable liquids. Maintain proper documentation of quantities and disposal methods for regulatory compliance.
Replace with Fresh Stock
Always use fresh, certified non-denatured ethanol to ensure consistent quality, regulatory compliance, and effectiveness in food, pharmaceutical, extraction, and analytical applications. When selecting replacement material:
Verify all required certifications (USP, FCC, organic, kosher, halal) on replacement stock before purchasing. Ensure the grade and purity level match your specific application requirements.
Source from reputable suppliers who provide comprehensive documentation, including certificates of analysis with current dating and complete traceability information.
Inspect packaging integrity upon delivery, ensuring that seals remain intact and that storage recommendations align with your facility capabilities.
Documentation and Traceability
Maintain proper records of disposal and replacement for regulatory compliance, particularly important for food and pharmaceutical applications requiring full traceability. Documentation should include:
-
Date and quantity of expired material disposed of
-
Disposal method and service provider used
-
Replacement material details, including lot numbers and certifications
-
Any testing performed on expired or replacement material
-
Investigation of root causes if expiration occurred earlier than expected
This documentation supports regulatory audits and helps identify opportunities to improve storage practices and reduce waste.
Proper Storage Extends Shelf Life
Implementing correct storage practices from the beginning maximizes shelf life and maintains regulatory compliance throughout the storage period.
Container Requirements
Keep non-denatured ethanol in appropriate food-grade, non-reactive containers that maintain regulatory compliance. Use containers specifically approved for food contact and beverage alcohol storage, such as glass, HDPE plastic, or stainless steel.
Original manufacturer packaging typically provides optimal protection until first use. When transferring to secondary containers, ensure they meet the same material compatibility and food-grade standards.
Temperature Control
Maintain cool storage temperatures at 10-25°C (50-77°F) following USP controlled room temperature guidelines and food processing facility standards. Avoid locations near heat sources, direct sunlight, or areas subject to temperature fluctuations.
Consider climate control systems in facilities located in hot climates or areas with significant seasonal temperature variations. The investment in temperature control prevents premature expiration and maintains product quality.
Light and Heat Protection
Protect from light and heat sources that could cause degradation and compromise food-grade quality. Use amber glass or opaque containers when possible, particularly for long-term storage applications.
Store containers away from windows and artificial lighting sources that emit UV radiation. Even fluorescent lighting can contribute to degradation over extended periods.
Proper Sealing
Ensure proper sealing to prevent evaporation, maintain concentration accuracy, and preserve regulatory compliance for food and pharmaceutical applications. Check seals regularly, particularly after each use, to verify integrity.
Consider using containers with positive-seal closures that provide visual confirmation of proper sealing. For frequently accessed containers, implement procedures to minimize opening time and exposure to ambient air.
Compliant Storage Facilities
Store in approved facilities meeting food processing and beverage alcohol storage requirements with adequate ventilation and fire safety systems. Facilities should comply with:
-
Local fire codes for flammable liquid storage
-
TTB regulations for beverage alcohol storage when applicable
-
FDA requirements for food-grade material storage
-
Any relevant organic, kosher, or halal storage standards
Regular Quality Monitoring
Implement regular testing protocols to verify continued compliance with USP, FCC, and other regulatory standards throughout the storage period. Testing frequency should be based on:
-
Application criticality and regulatory requirements
-
Storage conditions and their stability
-
Historical data on product performance
-
Proximity to manufacturer expiration dates
Maintain complete records of all quality monitoring activities to demonstrate ongoing compliance and identify trends that might indicate emerging storage problems.
Conclusion
Non-denatured ethanol generally lasts 3 years from manufacture for pure grades with excellent stability while maintaining food-grade and pharmaceutical compliance, though it can remain viable indefinitely under proper storage conditions. USP/FCC and organic certified formulations maintain the same 3-year shelf life, while diluted solutions like 140 proof have shorter shelf lives of approximately 2 years due to increased water content affecting stability.
The primary concerns are evaporation leading to concentration changes that affect regulatory compliance, light-induced degradation compromising food-grade quality, and maintaining traceability for food safety requirements. Proper storage in sealed, food-grade containers away from heat, light, and ignition sources is essential for maintaining quality, regulatory compliance, and maximizing the shelf life of this highly stable, food-safe, and versatile solvent.
For more detailed guidance on non-denatured ethanol handling and storage, consult your supplier's technical documentation, including Safety Data Sheets and certificates of analysis specific to your product grade and certification requirements.
For more detailed guidance, explore our Resource Library for comprehensive storage and handling information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Non-Denatured Ethanol You Can Trust?
If you are looking for high-quality non-denatured ethanol for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's non-denatured ethanol is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.









