Ethanol Safety & Hazards: Essential Handling Guidelines
Ethanol is a flammable organic solvent that is generally recognized as safe (GRAS) for food applications but poses significant fire hazards and can cause health effects through inhalation, skin contact, and ingestion. While approved for consumption in alcoholic beverages, industrial concentrations require careful handling due to flammability, vapor inhalation risks, and potential for intoxication.
Professionals working in food manufacturing, pharmaceuticals, cosmetics, laboratories, and industrial facilities must thoroughly understand the safety profile of ethanol. Despite its presence in everyday consumer products and natural production through fermentation, concentrated solutions and large volumes demand rigorous safety protocols to prevent fires and injuries.
Ethanol vapors are highly flammable and can cause intoxication through inhalation, making protective equipment and strict fire prevention measures critical. This guide provides essential information on ethanol safety, including identifying exposure symptoms and executing proper emergency responses.


In this article, we’ll explore:
- Chemical Hazards Overview
- Exposure Risks & Health Effects
- First Aid Measures
- Handling & Storage Precautions
- Spill & Leak Procedures
- Fire & Explosion Hazards
- Regulatory & Compliance Information
- Specialized Ethanol Types: Safety Considerations
- Technical Documents & References
Chemical Hazards Overview
Ethanol's hazard profile centers primarily on fire risk rather than chemical toxicity. The compound's low flash point and explosive vapor formation pose dangers that require constant vigilance around ignition sources and effective vapor control.
Hazard Classification
The Globally Harmonized System (GHS) assigns ethanol two pictograms. A flame symbol (GHS02) warns of highly flammable liquid and vapor, representing the dominant risk. An exclamation mark (⚠️ GHS07) signals irritation and harmful exposure effects.
NFPA ratings for ethanol are: Health 2 (moderate hazard), Flammability 3 (serious fire hazard), and Reactivity 0 (stable).
Primary Risks
Main hazards from ethanol vary with concentration and volume:
-
Fire danger: Extremely flammable with a flash point near 13°C (55°F). Vapors create explosive air mixtures and travel to remote ignition points. Static electricity during transfers can trigger ignition.
-
Breathing vapors: Produces dizziness, drowsiness, headache, nausea, and airway irritation. Heavy concentrations trigger narcotic effects, breathing suppression, and loss of consciousness.
-
Touching skin: Extended or frequent contact produces drying, irritation, and dermatitis. The compound penetrates skin substantially. Causes stinging on damaged or delicate skin.
-
Eye contact: Triggers substantial irritation with redness, watering, pain, and vision problems. Vapor contact also irritates the eyes.
-
Swallowing: Food-grade varieties are safe in typical beverage quantities. Yet substantial amounts or strong concentrations produce intoxication, nausea, vomiting, breathing suppression, coma, and possible death. Industrial versions may include poisonous denaturants.
Exposure Risks & Health Effects
Recognizing how ethanol affects health enables you to spot dangerous conditions and respond appropriately. Symptom intensity relates to concentration, duration, and entry method.
Immediate effects differ by exposure route. Breathing vapors creates quick effects like dizziness, headache, and drowsiness, with heavy concentrations causing stronger narcotic responses and breathing problems. Touching causes immediate drying and irritation with prolonged contact, and considerable absorption occurs through intact skin. Eye contact triggers immediate irritation, watering, and pain that briefly reduces vision.
Long-term exposure creates more substantial worries than many workplace chemicals. Extended contact with strong concentrations may produce liver injury, nervous system effects, and possible addiction. Repeated skin contact results in ongoing dermatitis and barrier damage. Though ethanol isn't carcinogenic, regular alcohol use is linked to multiple serious health problems, including liver disease and elevated cancer risk.
First Aid Measures
Rapid and correct first aid decreases injury severity from ethanol contact. Fire prevention takes absolute priority in any ethanol situation, with medical response following after controlling fire dangers.
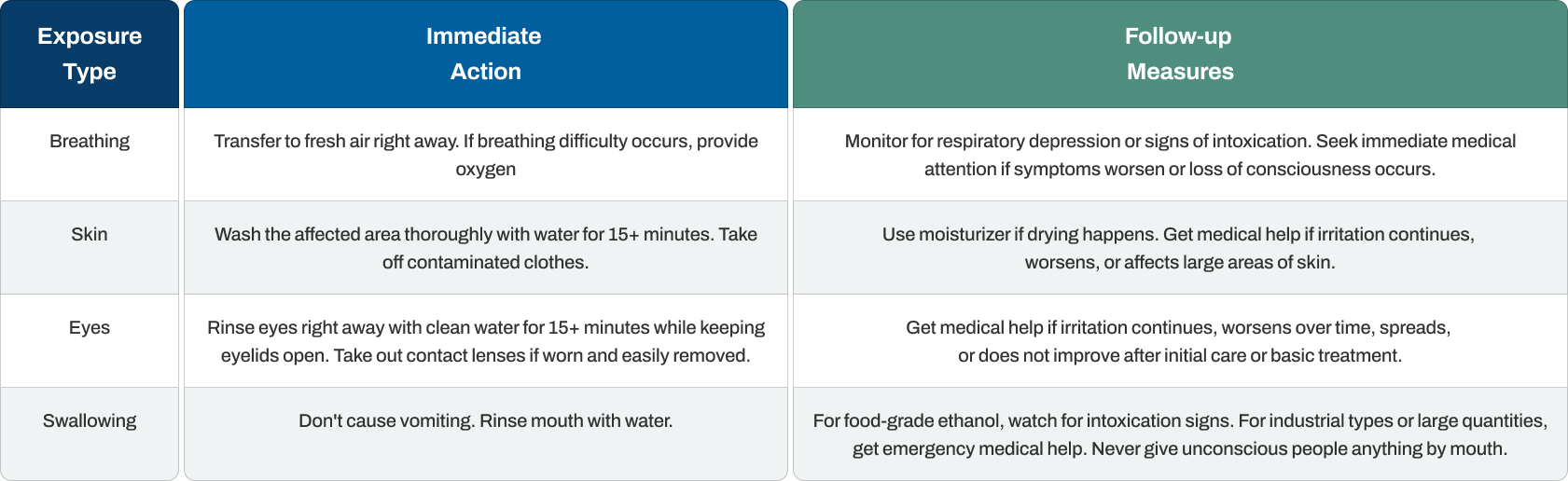
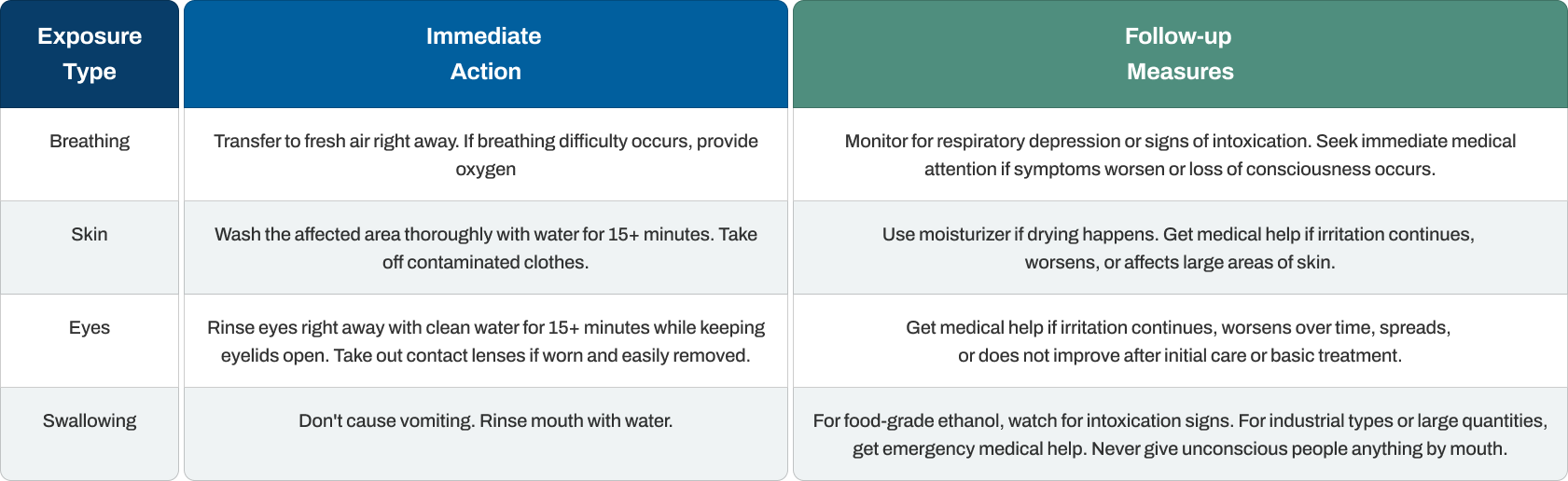
Key points include spotting intoxication indicators (unclear speech, poor coordination, confusion) that may appear after contact. Timing matters especially with breathing cases where respiratory suppression develops quickly. For swallowing industrial types with denaturants, emergency care is vital since denaturants like methanol produce blindness and death.
Handling & Storage Precautions
Working safely with ethanol demands systematic methods prioritizing fire prevention first, with contact prevention secondary.
Safe Handling Procedures
Wear chemical-resistant gloves (nitrile works well) when using ethanol to stop skin absorption and drying. Put on safety goggles and protective clothes to block splashes. Maintain strong ventilation to stop vapor buildup and keep levels below explosive ranges.
Remove all ignition possibilities, including open flames, hot areas, sparks, and static electricity. Install explosion-proof electrical gear where ethanol vapors might exist. Ground and bond containers during transfers to stop static discharge. Prevent static electricity by avoiding rapid pouring or splashing.
Don't eat, drink, or smoke where ethanol is used. Wash your hands completely after any contact. Keep protective gear properly and check frequently for wear.
Storage Requirements
Keep ethanol in cool, dry, well-ventilated spaces away from heat, sparks, flames, and oxidizing materials. Seal containers tightly when not using them to stop vapor release and evaporation. Place in approved flammable liquid cabinets or designated flammable areas meeting fire codes.
Keep storage temperature under 25°C to lower vapor pressure and fire danger. Shield from direct sun and heat sources. Apply proper grounding and bonding for all containers and transfer gear. Limit quantities to operational needs, placing excess in proper flammable facilities.
Frequent checks of storage spaces help find potential fire dangers before problems occur. Look for container damage, correct labels, sufficient ventilation, and removal of ignition sources. Confirm fire suppression systems work correctly.
Incompatible Substances
Ethanol reacts violently with powerful oxidizing agents, including permanganates, peroxides, and nitrates, which can ignite ethanol or trigger explosive reactions. Separate from powerful acids that may produce violent reactions when combined. Don't contact alkali metals (sodium, potassium), which react violently, creating flammable hydrogen gas.
Acetyl chloride and chromium trioxide react dangerously with ethanol. Don't contact hot surfaces and all ignition sources. Certain plastics and rubber materials are incompatible, breaking down or dissolving on contact.
Spill & Leak Procedures
Ethanol spills generate immediate fire dangers requiring quick action to stop ignition and manage vapors.
Emergency Response Steps
Remove all ignition sources right away, including electrical gear, open flames, smoking materials, and potential spark sources. Maintain good ventilation in the affected space, using explosion-proof fans if needed. Put on suitable protective gear, including flame-resistant clothes, chemical-resistant gloves, and safety goggles. Clear the area if a big spill happens, creating a safe boundary. Apply explosion-proof equipment only for cleanup work.
Containment Methods
Stop the spill from spreading using spill barriers or absorbent dikes. Absorb with inert, non-burning absorbent material like vermiculite or commercial spill products made for flammable liquids. Don't use sawdust or other burning absorbents that could add fire dangers. Dike big spills to stop flow into nearby areas.
Stop entry into waterways, sewers, basements, or tight spaces where vapors can gather and create explosion dangers. Vapor suppression foam may work for big spills to reduce vapor creation.
Proper Disposal
Collect spilled material where feasible for reuse or correct disposal. Move collected ethanol to suitable containers for recycling or waste disposal. Throw away contaminated absorbents as hazardous waste per local rules. Burn in an approved facility with sufficient scrubbing and energy recovery systems.
Don't release ethanol to waterways or sewers without the correct permits and treatment. Record spill events, including estimated amounts, cleanup methods used, and disposal steps followed for regulatory needs and improvement.
Fire & Explosion Hazards
Fire and explosion create the most serious dangers connected with ethanol use. Grasping fire behavior and correct response steps can stop injuries and deaths.
Flammability
Ethanol is extremely flammable with a flash point near 13°C (55°F), meaning it can ignite at relatively cool temperatures. It creates explosive vapor-air combinations across a broad concentration range. Vapors are heavier than air and may move considerable distances to find ignition sources, then flash back to the source.
The autoignition temperature is roughly 365°C (689°F). Under this temperature, an ignition source is needed, but above it, ethanol vapors will spontaneously ignite without outside spark or flame.
Extinguishing Media
Apply alcohol-resistant foam specifically made for polar solvent fires. Regular firefighting foams fail on contact with ethanol and don't work. Dry chemical extinguishers work for small fires. Carbon dioxide works for electrical fires involving ethanol. Water spray can cool containers and dilute spills, but don't use straight water streams, which may spread burning liquid.
Water on burning ethanol creates steam that can splash burning liquid. Yet water stays valuable for cooling nearby containers and structures to stop fire spread.
Firefighting Precautions
Apply respiratory protection with supplied air and full protective gear, including flame-resistant clothes. Approach fires from upwind to prevent smoke and vapor breathing. Cool containers with water spray from maximum distance to stop rupture from heat contact.
Ethanol burns with a nearly invisible flame in daylight, making fire detection hard. Know that vapors may reignite after apparent extinguishment. Tank fires create particular dangers, including potential boilover, where water trapped under burning ethanol suddenly boils, throwing burning liquid from the tank.
Regulatory & Compliance Information
Ethanol faces extensive regulatory requirements varying by application, concentration, and planned use. Grasping these regulations helps maintain compliance and promotes safe operations.
OSHA Regulations
Ethanol is regulated as a flammable liquid needing compliance with 29 CFR 1910.106 (flammable and combustible liquids standard). The permissible exposure limit (PEL) is 1000 ppm (1900 mg/m³) as an 8-hour time-weighted average. Operations must implement engineering controls, supply suitable personal protective gear, and train workers on fire dangers.
Flame-resistant clothes are needed in hazardous areas where fire or explosion risks exist. Explosion-proof electrical gear must be installed in classified locations. Emergency eyewash stations and safety showers are needed where eye or skin contact may happen.
EPA & Environmental Impact
Ethanol breaks down readily with low buildup potential in water systems. Yet it may harm aquatic life in large quantities through oxygen loss as it degrades. Releases exceeding the reportable quantity under CERCLA must be reported to the National Response Center.
Air releases from ethanol use may face state and local rules, particularly in areas with air quality worries. Operations should implement correct vapor recovery systems to reduce environmental releases.
DOT Transportation Regulations
Ethanol is classified as Hazardous Material Class 3 (Flammable Liquid) for transport purposes. UN1170 applies to ethanol solutions above 24% by volume. Shipments need correct shipping names, hazard labels, placards for bulk amounts, and packaging meeting DOT specifications.
Special provisions apply for fuel ethanol transport. Drivers moving bulk amounts must have suitable hazmat endorsements. Shipping papers must accurately describe the material and amount.
FDA/USP/ACS Compliance
Food-grade ethanol has GRAS status for food applications under FDA rules, and beverages are regulated by the Alcohol and Tobacco Tax and Trade Bureau (TTB) with strict controls on production, distribution, and taxation. Industrial ethanol may need permits depending on use and amount.
Denatured alcohol is exempt from beverage alcohol taxes but regulated for industrial use. Specific denaturant formulas are approved for different applications. Operations using denatured alcohol must comply with TTB rules regarding storage, use records, and reporting.
Specialized Ethanol Types: Safety Considerations
Different ethanol grades create unique safety considerations beyond standard handling steps. Grasping these differences helps promote suitable precautions for specific products.
Denatured Alcohol
Denatured alcohol includes poisonous denaturants, including methanol, isopropanol, or other substances, making it unsuitable for drinking. These additives create additional health dangers beyond ethanol itself. Swallowing may produce blindness, organ damage, or death depending on the denaturant type and quantity consumed.
Handle denatured alcohol with extra precautions due to denaturant toxicity. Never apply denatured alcohol in food applications or where human consumption could happen. Clearly mark containers to stop accidental drinking. Review the specific denaturant formula to grasp additional dangers.
Absolute Ethanol (200 Proof)
Absolute ethanol is extremely hygroscopic, quickly absorbing moisture from the air. This quality creates handling challenges as contact with atmospheric moisture immediately begins dilution. It creates a higher fire risk due to the absence of water content that would moderate flammability.
Keep absolute ethanol under nitrogen or other inert atmosphere to stop moisture absorption. Apply quickly after opening containers, as water absorption is rapid. The absence of water content may produce more severe dehydration effects on skin and mucous membranes compared to dilute ethanol.
Food Grade Ethanol
Food-grade ethanol is safe for drinking in moderate quantities as found in alcoholic beverages. Yet it still creates serious fire dangers, needing the same fire prevention measures as industrial grades. Handle using food-grade steps to stop contamination while keeping strict fire safety protocols.
Concentrations above certain thresholds become subject to beverage alcohol rules even in food-grade applications. Operations must comply with applicable TTB regulations and keep proper permits.
Laboratory Grade
Laboratory-grade ethanol may include impurities or stabilizers depending on the specific grade and planned analytical use. Follow the specific Safety Data Sheet for each grade as dangers may vary. Typically needs laboratory safety protocols, including fume hoods for vapor control and suitable chemical storage.
Extra health dangers may exist depending on purity specifications and any additives present. Reagent grades generally have fewer impurities but keep the same fire dangers as technical grades.
Technical Documents & References
Working safely with ethanol needs access to current and accurate technical documentation addressing both fire dangers and health effects.
Safety Data Sheets (SDS)
Safety Data Sheets from suppliers give detailed safety information, handling steps, and first aid measures specific to concentration, grade, and denaturant formula. These documents are legally needed under OSHA's Hazard Communication Standard and must be readily available to all workers.
Review SDS documents before starting work with ethanol and update when new versions become available. Pay particular attention to sections covering fire dangers, exposure limits, and emergency steps.
TTB Regulations
Alcohol and Tobacco Tax and Trade Bureau regulations for beverage and industrial alcohol give compliance requirements for permits, labeling, record-keeping, and handling steps. These regulations are complex and vary depending on alcohol use and concentration.
Operations using ethanol must determine which TTB regulations apply to their work and keep suitable permits. Consult TTB guidance documents or qualified compliance specialists for specific situations.
Certificates of Analysis (COA)
Certificates of Analysis confirm product purity, water content, denaturant type and concentration, and compliance with food, pharmaceutical, or technical standards. These documents verify specific batches meet the needed specifications and give traceability for quality control.
COAs are particularly important for food-grade and pharmaceutical applications where impurity levels must meet strict specifications. Request and keep COAs for all incoming ethanol shipments to document product quality and regulatory compliance.
Final Note
Ethanol is valuable yet hazardous, demanding strict fire prevention measures and correct safety steps. Though its toxicity is relatively low compared to many workplace chemicals, its extreme flammability creates serious risks that have caused numerous workplace fires, injuries, and deaths.
Success with ethanol safety comes from consistently removing ignition sources, maintaining proper ventilation to manage vapors, having suitable fire suppression gear, and providing thorough worker training. By following established fire safety protocols and recognizing concentration-dependent health dangers, you can reduce risks while safely applying this versatile chemical across food, pharmaceutical, laboratory, and industrial applications.
Explore our Resource Library for more information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Ethanol You Can Trust?
If you are looking for high-quality ethanol for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's ethanol is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.









