Lactic Acid Safety & Hazards: Essential Handling Guidelines
Lactic acid is an organic acid that is generally recognized as safe (GRAS) for food applications but can irritate the skin, eyes, and respiratory system in concentrated forms. While not classified as a carcinogen and considered relatively safe compared to other acids, it is mildly corrosive in concentrated solutions and may cause burns with prolonged exposure.
Understanding lactic acid's safety profile is important for professionals across food manufacturing, cosmetics, pharmaceuticals, and industrial settings. Despite its natural occurrence in fermented foods and widespread use in consumer products, concentrated lactic acid solutions demand respect and proper handling procedures to prevent injuries.
Since concentrated lactic acid can cause respiratory irritation, skin burns, and serious eye damage, proper protective equipment and handling procedures are essential. This comprehensive guide covers everything you need to know about lactic acid safety, from recognizing exposure symptoms to implementing proper emergency procedures.


In this article, we’ll explore:
- Chemical Hazards Overview
- Exposure Risks & Health Effects
- First Aid Measures
- Handling & Storage Precautions
- Spill & Leak Procedures
- Fire & Explosion Hazards
- Regulatory & Compliance Information
- Technical Documents & References
Chemical Hazards Overview
Lactic acid presents a mild to moderate hazard profile that requires attention to safety protocols, particularly when working with concentrated solutions. Unlike highly reactive chemicals, lactic acid is stable under normal conditions, but its corrosive properties at higher concentrations make proper handling essential.
Hazard Classification
Under the Globally Harmonized System (GHS), lactic acid displays two pictograms.
- The exclamation mark (⚠️ GHS07) indicates irritation and harmful effects.
- The corrosion symbol (⚗️ GHS05) appears for concentrated solutions, indicating corrosive effects on skin and eyes.
The NFPA hazard rating system assigns lactic acid: Health 1-2 (slight to moderate hazard), Flammability 1 (slight fire hazard), and Reactivity 0 (stable).
Primary Risks
The main dangers from lactic acid exposure depend significantly on concentration:
-
Respiratory irritation: Inhalation of vapors from concentrated or heated solutions can irritate the respiratory tract. Generally well-tolerated at typical use concentrations.
-
Skin contact: Concentrated solutions (>10%) may cause irritation, redness, and chemical burns with prolonged contact. Dilute solutions are generally well-tolerated.
-
Eye exposure: Can cause eye burns and serious damage. Symptoms include severe redness, pain, tearing, potential corneal damage, and chemical conjunctivitis.
-
Ingestion: GRAS for food applications. However, large amounts of concentrated lactic acid can cause nausea, vomiting, abdominal pain, and gastrointestinal irritation.
Exposure Risks & Health Effects
Understanding how lactic acid affects the human body helps you recognize potentially dangerous situations. The severity of symptoms depends largely on the concentration and duration of exposure.
Acute exposure effects vary by concentration. Respiratory symptoms from vapors include coughing and throat discomfort, though uncommon with dilute solutions at room temperature. Skin contact with concentrated solutions (>10%) produces irritation and possible burns, while dilute solutions used in cosmetics are well-tolerated. Eye exposure causes immediate pain, tearing, and potential corneal damage requiring prompt treatment.
Chronic exposure presents minimal concerns. Lactic acid is not classified as carcinogenic and is generally safe for long-term exposure at typical use levels. Its GRAS status indicates safety for regular consumption in foods, distinguishing it from many industrial chemicals that present cumulative health risks.
First Aid Measures
A quick and appropriate first aid response can significantly reduce the severity of lactic acid exposure injuries. Immediate action remains the priority for all exposure routes.
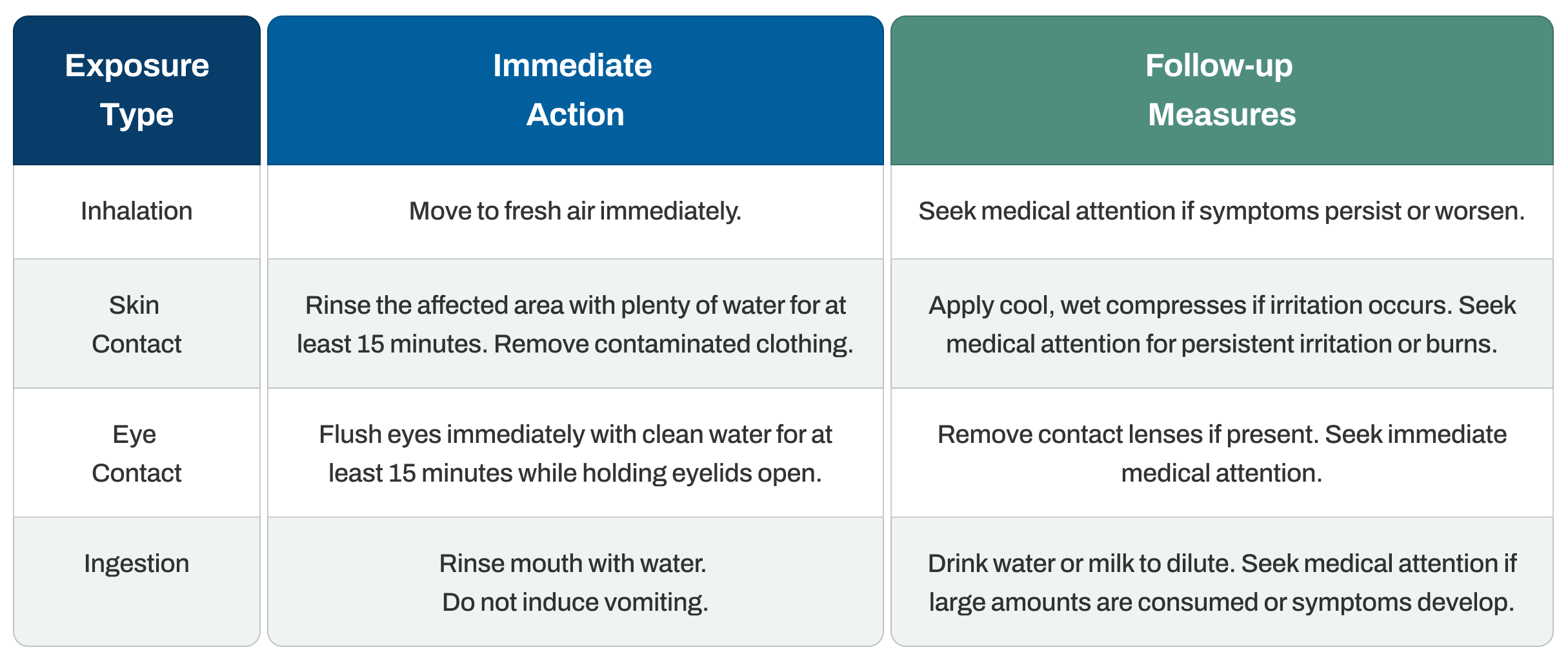
Critical considerations include using clean water for all rinsing procedures and avoiding self-contamination while helping others. Time is particularly important with eye exposure, as delayed treatment can result in corneal damage. For ingestion, never induce vomiting unless directed by poison control, as this could cause additional esophageal damage.
Handling & Storage Precautions
Safe handling of lactic acid requires a systematic approach that addresses both immediate safety needs and long-term exposure prevention.
Safe Handling Procedures
Use chemical-resistant gloves (nitrile or neoprene) when handling concentrated solutions. Wear safety goggles and protective clothing to prevent skin and eye contact. Ensure adequate ventilation, particularly when heating solutions or working with large quantities.
Avoid generating mists or vapors through vigorous mixing or heating without proper controls. Never eat, drink, or smoke in areas where lactic acid is being handled. Wash hands thoroughly after any contact and remove contaminated clothing before leaving work areas.
Storage Requirements
Store lactic acid in cool, dry, well-ventilated areas at 15-25°C. Keep containers tightly closed to prevent moisture absorption and evaporation. Protect from excessive heat and direct sunlight. Use original labeled containers whenever possible, and ensure secondary containment for larger quantities.
Regular inspection of storage areas helps identify potential problems before they become serious incidents. Check for container damage, proper labeling, and adequate ventilation systems.
Incompatible Substances
Lactic acid reacts with strong oxidizing agents (peroxides, permanganates, nitrates), which can lead to violent reactions. Keep it separated from strong bases (sodium hydroxide, potassium hydroxide), which react exothermically, generating heat. Avoid contact with certain metals (aluminum, zinc) and carbonates/bicarbonates, which can cause gas evolution.
Spill & Leak Procedures
Lactic acid spills require prompt attention to prevent exposure and environmental damage.
Emergency Response Steps
Ensure adequate ventilation in the affected area. Wear appropriate protective equipment, including chemical-resistant gloves, safety goggles, and protective clothing. Evacuate non-essential personnel and contain the spill to prevent spreading.
Containment Methods
Absorb small spills with inert absorbent materials such as vermiculite, sand, or commercial spill control products. Neutralize with sodium bicarbonate or lime before cleanup, adding neutralizing agent gradually. Monitor pH during neutralization to ensure complete reaction. Flush the area with water after neutralization.
Proper Disposal
Neutralize lactic acid before disposal according to local regulations. The compound is biodegradable and can be treated in wastewater systems after neutralization, making disposal simpler than with many industrial acids. Avoid direct release of concentrated solutions to the environment.
Document spill incidents, including estimated quantities, cleanup methods used, and disposal procedures followed. This information supports regulatory compliance and helps improve future response procedures.
Fire & Explosion Hazards
While lactic acid presents relatively low fire hazards, understanding its behavior in fire situations helps ensure appropriate emergency response.
Flammability
Lactic acid is a combustible liquid with a flash point around 122°C (252°F). This relatively high flash point means it is not easily ignited under normal conditions. The compound can burn when heated above its flash point or when involved in fires started by other materials.
Extinguishing Media
Water spray, dry chemical, carbon dioxide, or alcohol-resistant foam can effectively extinguish lactic acid fires. Water is particularly effective and can be used to cool containers and dilute spills.
Firefighting Precautions
Use respiratory protection and full protective gear when fighting fires involving lactic acid. Avoid inhalation of combustion products, which may include carbon monoxide and carbon dioxide. Water used to fight fires may become acidic and may require pH adjustment before discharge.
Regulatory & Compliance Information
Lactic acid is subject to various regulatory requirements that vary by application and concentration. Understanding these regulations helps maintain compliance and ensures safe use.
OSHA Regulations
Lactic acid requires standard chemical handling precautions and appropriate PPE use. It generally receives a lower hazard classification than many industrial acids due to its mild corrosivity. Employers must provide appropriate training on safe handling procedures for workers who may be exposed.
While not subject to specific OSHA permissible exposure limits, general chemical safety standards apply to prevent skin, eye, and respiratory irritation. Maintain Safety Data Sheets and ensure workers have access to hazard information.
EPA & Environmental Impact
Lactic acid presents a low environmental hazard. The compound is biodegradable and environmentally acceptable, breaking down rapidly in natural systems. Minimal aquatic toxicity occurs at typical use concentrations, though concentrated releases should be prevented to avoid temporary pH alterations.
Facilities should implement proper containment measures to prevent accidental releases to waterways. Environmental remediation following lactic acid releases is generally straightforward due to its biodegradability and low toxicity.
DOT Transportation Regulations
Lactic acid is generally not classified as a hazardous material for transportation unless shipped at very high concentrations. Most commercial formulations can be transported using standard chemical shipping procedures without special hazardous materials documentation. Follow standard chemical shipping procedures, including proper labeling, appropriate packaging to prevent leaks, and documentation identifying the material.
FDA/USP/ACS Compliance
GRAS status for food applications confirms lactic acid's safety for use in foods and beverages at appropriate levels. This FDA designation reflects extensive safety data and decades of safe use. USP grade is available for pharmaceutical applications, meeting strict purity standards. FCC grade ensures safety and purity standards for food applications, with limits on heavy metals and impurities. These certifications require comprehensive quality control and testing to verify compliance with applicable standards. Certificates of analysis document that specific batches meet required specifications.
Technical Documents & References
Working with lactic acid safely requires access to current and accurate technical documentation.
Safety Data Sheets (SDS)
GRAS status for food applications confirms lactic acid's safety for use in foods and beverages at appropriate levels. This FDA designation reflects extensive safety data and decades of safe use. USP grade is available for pharmaceutical applications, meeting strict purity standards. FCC grade ensures safety and purity standards for food applications, with limits on heavy metals and impurities. These certifications require comprehensive quality control and testing to verify compliance with applicable standards. Certificates of analysis document that specific batches meet required specifications.
GRAS Determination Documents
GRAS determination documents and toxicological studies support safety for food and cosmetic applications. These studies demonstrate lactic acid's safety profile through historical use data and modern toxicological testing.
Review these documents when evaluating lactic acid for new food or cosmetic applications to ensure your intended use aligns with established safety parameters.
Certificates of Analysis (COA)
Certificates of Analysis verify product purity, pH, and compliance with food, pharmaceutical, or technical standards. These documents confirm specific batches meet required specifications and provide traceability.
COAs are particularly important for pharmaceutical and food-grade applications where impurity levels must meet strict specifications. Request and retain COAs for all incoming lactic acid shipments to document product quality.
Laboratories and facilities using lactic acid should maintain easy access to these documents and ensure all personnel receive proper safety training. Emergency contact information for poison control centers should be readily available.
Final Note
Lactic acid is a relatively safe chemical that still demands careful handling and proper safety procedures, particularly when working with concentrated solutions. While significantly less hazardous than many industrial acids, it requires respect and systematic management to prevent injuries.
Success in lactic acid safety comes from consistent application of proper procedures, appropriate protective equipment use, and maintaining current technical documentation. By following established protocols and understanding the compound's concentration-dependent hazards, you can minimize risks while safely utilizing this versatile chemical across food, cosmetic, pharmaceutical, and industrial applications.
Explore our Resource Library for more information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Lactic Acid You Can Trust?
If you are looking for high-quality lactic acid for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's lactic acid is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.








