Non-Denatured Ethanol Safety & Hazards: A Complete Guide
When you work with non-denatured ethanol, you're handling a substance that walks an interesting line. It's safe enough to drink in your evening glass of wine, yet concentrated enough in industrial settings to pose serious fire risks and health hazards. Understanding this dual nature is essential for anyone who handles, stores, or works around non-denatured ethanol in any capacity.
Unlike its denatured counterpart, non-denatured ethanol doesn't contain toxic additives that make it unfit for consumption. This makes it valuable for food production, pharmaceuticals, and beverage manufacturing. However, this purity doesn't make it any less dangerous when it comes to flammability or the risks associated with concentrated exposure.
The key difference lies in what's missing rather than what's present. Denatured alcohol contains poisonous substances specifically added to make it undrinkable. Non-denatured ethanol is pure ethyl alcohol, which explains why it carries a Generally Recognized as Safe (GRAS) status from the FDA for food applications. Yet this same purity means it requires stricter regulatory oversight, particularly from the Alcohol and Tobacco Tax and Trade Bureau (TTB).


In this article, we’ll explore:
- Chemical Hazards Overview
- Exposure Risks & Health Effects
- First Aid Measures
- Handling & Storage Precautions
- Spill & Leak Procedures
- Fire & Explosion Hazards
- Regulatory & Compliance Information
- Specialized Non-Denatured Ethanol Types: Safety Considerations
- Technical Documents & References
Chemical Hazards Overview
Non-denatured ethanol presents a hazard profile dominated by its extreme flammability, though its effects on human health also warrant serious attention. The absence of toxic denaturants actually simplifies the safety picture in some ways, as you don't need to worry about methanol poisoning or the additional irritants found in denatured formulations.
Hazard Classification
The Globally Harmonized System assigns non-denatured ethanol two distinct pictograms that tell you everything about its primary risks. The flame symbol (GHS02) marks it as a highly flammable liquid and vapor, while the exclamation mark (⚠️ GHS07) indicates harmful and irritating effects.
Under the NFPA hazard rating system, non-denatured ethanol receives a Health rating of 2, indicating moderate hazard potential. The Flammability rating sits at 3, signaling a serious fire hazard that requires immediate attention in any safety planning. Reactivity gets a 0, meaning the compound remains stable under normal conditions.
Primary Risks
The dangers from non-denatured ethanol come through multiple pathways, each presenting unique challenges for workplace safety:
-
Extreme flammability: With a flash point around 13°C (55°F), non-denatured ethanol can ignite at surprisingly ordinary temperatures. Vapors are heavier than air and travel to distant ignition sources. Static electricity can easily ignite vapors during transfer operations.
-
Vapor inhalation: High concentrations cause intoxication, dizziness, respiratory depression, and potentially unconsciousness. Chronic exposure may affect the central nervous system and liver.
-
Skin absorption: Prolonged contact causes drying, irritation, and dermatitis. Ethanol absorbs through the skin in significant amounts, potentially causing systemic effects.
-
Eye irritation: Causes serious irritation with redness, tearing, pain, and temporary vision impairment. Pure ethanol is less irritating than denatured formulations.
-
Consumption considerations: Pure ethanol is safe for consumption in moderate amounts as found in alcoholic beverages, but large amounts or high concentrations can cause intoxication, respiratory depression, and potentially death.
-
Invisible flames: Burns with a nearly invisible flame in daylight, creating unique hazards for firefighters and emergency responders.
-
Hot surface hazards: Avoid contact with hot surfaces and ignition sources that can provide autoignition temperatures.
Exposure Risks & Health Effects
Understanding how non-denatured ethanol affects your body helps you recognize dangerous situations before they escalate into medical emergencies. The concentration and duration of exposure largely determine the severity of symptoms.
Acute exposure effects develop quickly after contact. Respiratory symptoms include dizziness, drowsiness, headache, and nausea when vapors are inhaled. Skin contact produces drying, irritation, and may cause a stinging sensation on broken or sensitive skin, though it lacks the toxic denaturants found in denatured alcohol. Eye exposure causes immediate pain, tearing, and potential vision problems.
Ingestion presents a unique situation with non-denatured ethanol. In moderate amounts, as found in alcoholic beverages and food applications, it's perfectly safe due to its GRAS status. However, large amounts or high concentrations of industrial-grade material can cause severe intoxication, nausea, vomiting, respiratory depression, coma, and potentially death. Unlike denatured alcohol, it does not contain toxic denaturants.
Chronic exposure concerns arise from repeated contact over extended periods. Long-term exposure can lead to liver damage, neurological effects, and persistent respiratory irritation. Workers who handle non-denatured ethanol regularly without proper protection may develop chronic dermatitis and skin sensitization. While not classified as carcinogenic, chronic excessive alcohol consumption is associated with various health risks, including liver disease and increased cancer risk.
First Aid Measures
Responding quickly and appropriately to non-denatured ethanol exposure can dramatically reduce the severity of injuries and long-term health consequences. The specific steps you take depend on how exposure occurred, but immediate action remains the common priority.
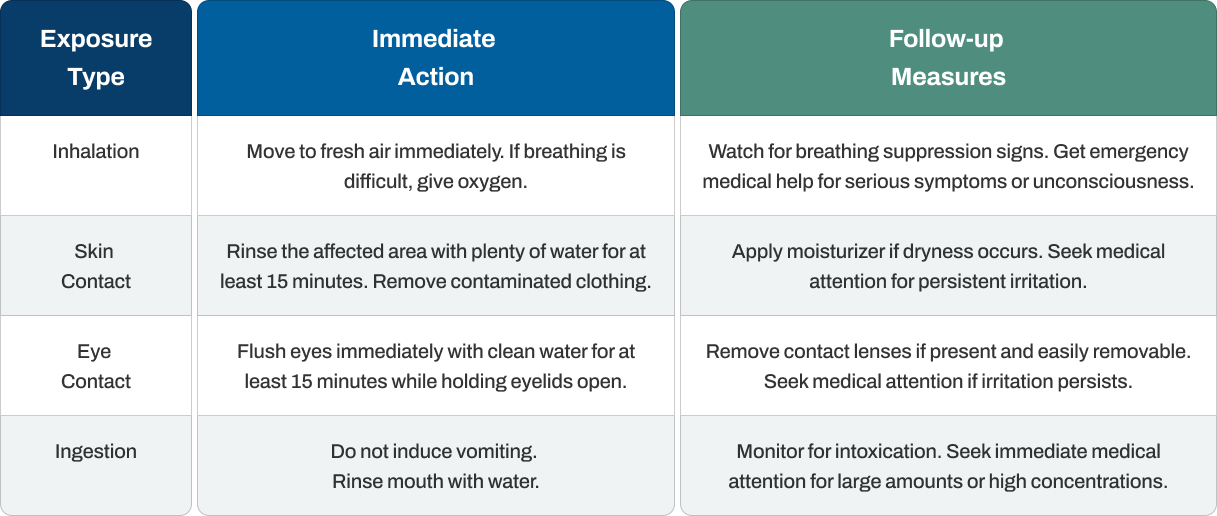
Critical considerations include avoiding contamination of yourself while helping others. Time matters enormously with eye exposure, as damage can occur within minutes of contact. The absence of toxic denaturants in non-denatured ethanol simplifies medical response compared to denatured alcohol exposures.
Handling & Storage Precautions
Safe management of non-denatured ethanol requires systematic attention to both fire prevention and health protection. The combination of extreme flammability and absorption risks means your safety procedures must address multiple hazard categories simultaneously.
Safe Handling Procedures
Personal protective equipment forms your first line of defense. Always use chemical-resistant gloves, preferably nitrile, as standard latex or vinyl gloves degrade quickly upon contact with ethanol. Wear safety goggles and protective clothing to prevent skin contact and eye exposure.
Ensure excellent ventilation in all work areas. Local exhaust ventilation at transfer points captures vapors at their source, while general area ventilation prevents accumulation in low-lying areas where ethanol vapors tend to collect.
Eliminate all ignition sources before handling non-denatured ethanol. Use explosion-proof electrical equipment, ground and bond containers during transfer operations, and avoid generating static electricity through proper procedures and conductive footwear. Handle with the same fire precautions as denatured ethanol, avoiding hot surfaces and any potential ignition sources.
Storage Requirements
Store non-denatured ethanol in cool, dry, well-ventilated areas away from heat, sparks, open flames, and oxidizing materials. Keep containers tightly closed and maintain temperatures below 25°C (77°F). Use approved flammable liquid storage cabinets that include spark-proof ventilation and fire-resistant construction.
Protect containers from direct sunlight, which can heat them significantly above ambient temperature and increase vapor pressure. Ensure proper grounding and bonding provisions are in place. If storing beverage alcohol, ensure compliance with TTB storage regulations.
Incompatible Substances
Non-denatured ethanol must be kept separated from several dangerous materials:
-
Strong oxidizing agents: Permanganates, peroxides, and nitrates can react violently, causing immediate ignition or explosive reactions.
-
Strong acids: Generate heat when mixed with ethanol, creating vapor release and potential ignition scenarios.
-
Alkali metals: React violently with alcohols, releasing hydrogen gas and heat.
-
Specialty chemicals: Acetyl chloride, chromium trioxide, and platinum can react dangerously under various conditions.
-
Hot surfaces and ignition sources: Avoid contact with any heat sources that could reach autoignition temperatures.
Additionally, non-denatured ethanol is incompatible with some plastics and rubber materials, which can degrade upon contact. Always verify container material compatibility before use.
Spill & Leak Procedures
Non-denatured ethanol spills demand immediate attention because of the extreme fire hazard they create. A small spill can rapidly generate an explosive atmosphere, making response time critical. Pure ethanol spills are less toxic than denatured alcohol but equally flammable.
Emergency Response Steps
First, ensure the safety of all personnel in the area. Eliminate all ignition sources immediately by shutting down electrical equipment, extinguishing flames, and stopping operations that might generate sparks. Evacuate non-essential workers and establish proper ventilation.
All cleanup personnel must wear appropriate protective equipment, including flame-resistant clothing, respirators, chemical-resistant gloves, and eye protection. Use explosion-proof equipment only. Large spills may require evacuation of the entire area until vapors can be dispersed. Consider that alcohol vapors are heavier than air and will settle in low-lying areas.
Containment Methods
Use non-combustible absorbent materials such as inert absorbents, vermiculite, or diatomaceous earth. Do not use sawdust or other combustible absorbents, as they create additional fuel for potential fires. Dike large spills to prevent spreading.
Contain the spill to prevent entry into waterways, sewers, or confined spaces. Work carefully to minimize vapor generation. Use non-sparking tools throughout the cleanup process. For large spills, consider specialized equipment like vacuum trucks with explosion-proof motors.
Water spray can help knock down vapors and dilute the spill, though this creates additional liquid to manage. Alcohol-resistant foam provides better vapor suppression without as much dilution.
Environmental Protection and Disposal
Never allow non-denatured ethanol to enter storm drains, waterways, or soil. While less toxic than denatured alcohol due to the absence of poisonous additives, pure ethanol still causes environmental damage, particularly to aquatic ecosystems.
Recover spilled material where possible for reuse. Dispose of contaminated absorbents as hazardous waste according to local and TTB regulations. Incinerate in an approved facility with adequate scrubbing and energy recovery. Do not discharge to waterways. Follow beverage alcohol disposal regulations if applicable.
Document the spill incident thoroughly for regulatory compliance and future prevention.
Fire & Explosion Hazards
The fire and explosion characteristics of non-denatured ethanol dominate its hazard profile and drive most safety requirements. Understanding how ethanol burns and how to respond to ethanol fires can mean the difference between a minor incident and a catastrophic loss.
Flammability Characteristics
Non-denatured ethanol is a highly flammable liquid with a flash point around 13°C (55°F), meaning it forms explosive vapor-air mixtures at surprisingly low temperatures. Vapors are heavier than air and may travel to distant ignition sources. The autoignition temperature sits at approximately 365°C (689°F).
The substance burns with a nearly invisible flame in daylight, creating extreme danger for workers who may walk into burning areas without seeing the fire. Ethanol forms explosive mixtures with air across a broad concentration range, and vapors may reignite after initial suppression.
Extinguishing Methods
Alcohol-resistant foam is essential for effective fire suppression, as standard foams break down quickly on alcohol fires. Other suitable agents include dry chemical, carbon dioxide, or water spray. Do not use straight water streams, which may spread fire. Water can be used to cool containers and dilute spills in controlled applications.
Firefighting Precautions
Firefighters must use respiratory protection and full protective gear. Approach fires from upwind positions and cool containers with water spray to prevent rupture. Non-denatured ethanol burns with a nearly invisible flame in daylight, making thermal imaging cameras particularly valuable.
Be aware of potential boilover in tank fires and consider alcohol vapor density and dispersion patterns when establishing safety zones. Vapors may reignite even after flames appear extinguished, requiring continued monitoring and vapor suppression.
Regulatory & Compliance Information
Non-denatured ethanol faces more complex regulatory oversight than most industrial chemicals because of its dual nature as both a beverage ingredient and an industrial solvent. Multiple federal agencies exercise jurisdiction, creating compliance obligations that extend beyond standard chemical safety requirements.
OSHA Requirements
The Occupational Safety and Health Administration regulates non-denatured ethanol under 29 CFR 1910.106, the flammable liquids standard. The permissible exposure limit (PEL) is 1000 ppm (1900 mg/m³) as an 8-hour time-weighted average, identical to denatured ethanol.
Employers must provide appropriate training, personal protective equipment, and safety procedures. Flame-resistant clothing and explosion-proof equipment are required in hazardous areas. Hazard communication requirements mandate Safety Data Sheets, proper labeling, and worker training.
EPA and Environmental Regulations
The Environmental Protection Agency considers ethanol readily biodegradable with low bioaccumulation potential. It's generally less harmful to aquatic life than denatured alcohol due to the lack of toxic denaturants. Reportable quantity thresholds under CERCLA require notification when releases exceed specified amounts.
Air emissions regulations in some jurisdictions classify ethanol as a volatile organic compound subject to permitting and control requirements. Facilities must comply with regulations regarding wastewater discharge and environmental releases.
DOT Transportation Requirements
The Department of Transportation classifies non-denatured ethanol solutions containing more than 24% alcohol by volume as Hazardous Material Class 3 (Flammable Liquid), UN1170. This requires proper shipping names, labels, placards, and packaging specifications.
Shipping papers must include complete hazard information. Vehicle placarding becomes necessary when quantities exceed specific thresholds. Special provisions may apply for fuel ethanol shipments. Higher regulatory oversight applies compared to denatured alcohol.
FDA and TTB Compliance
Non-denatured ethanol has GRAS status for food applications, confirming safety for intended food and beverage uses. The TTB regulates beverage alcohol with strict permitting requirements for production, storage, and processing facilities.
Industrial non-denatured ethanol requires federal excise tax payment or special permits. Alcoholic beverages are subject to taxation above certain concentrations. USP (United States Pharmacopeia) and FCC (Food Chemicals Codex) grades meet pharmaceutical and food purity standards, requiring additional quality controls and documentation.
Specialized Non-Denatured Ethanol Types: Safety Considerations
Different grades and certifications of non-denatured ethanol share the same core hazards but require specific handling considerations based on their intended applications.
USP/Food Grade Ethanol meets pharmaceutical and food purity standards, making it safe for consumption and food applications. Despite this food-grade status, it still poses fire hazards identical to other ethanol concentrations. Handling requires food-grade procedures with stricter purity and contamination controls.
Absolute Ethanol (200 Proof) is extremely hygroscopic and flammable, absorbing moisture rapidly from the air. It requires special storage conditions to prevent water absorption. The lack of water content creates maximum flammability potential and may cause more severe dehydration effects on the skin.
Organic Certified Ethanol is produced without synthetic chemicals or denaturants and meets USDA organic standards. While safe for consumption in organic applications, it poses identical fire hazards and requires organic handling and storage procedures to maintain certification.
Kosher/Halal Certified Ethanol meets religious purity requirements for dietary applications. These certifications address religious compliance in production and handling but don't change the fundamental safety hazards. The same fire and safety precautions apply as with other non-denatured ethanol.
Technical Documents & References
Working with non-denatured ethanol safely requires access to current and accurate technical documentation that addresses its unique characteristics.
Safety Data Sheets (SDS) provide detailed safety, handling, and first aid measures specific to concentration, grade, and purity level without denaturant considerations. Multiple suppliers may offer varying levels of detail, so compare information when possible.
TTB Regulations for beverage and industrial alcohol provide compliance requirements for permits, taxation, labeling, and handling of non-denatured ethanol. These regulations are essential for facilities that produce, store, or process beverage-grade alcohol.
FDA GRAS Documentation confirms safety for food applications and consumption, with guidelines for acceptable use levels in food and beverage applications. This documentation supports the use of non-denatured ethanol in products intended for human consumption.
Certificate of Analysis (COA) verifies product purity, water content, absence of denaturants, and compliance with food, pharmaceutical, USP, FCC, or organic standards. These documents are essential for quality control and regulatory compliance.
Laboratories and facilities using non-denatured ethanol should maintain easy access to these documents and ensure all personnel receive proper safety training. Emergency contact information for poison control centers and environmental agencies should be readily available for quick expert assistance.
Final Note
Non-denatured ethanol occupies a unique position as both a food-safe ingredient and a highly hazardous industrial chemical. This dual nature requires you to maintain constant awareness of both its extreme flammability and its effects on human health.
The absence of toxic denaturants makes non-denatured ethanol safer in some respects than its denatured counterpart, particularly in cases of accidental ingestion or skin contact. However, this relative safety doesn't extend to fire hazards, where all types of ethanol pose identical serious risks.
Success in non-denatured ethanol safety comes from consistent application of proper procedures, regular training, and maintaining current technical documentation. By following established protocols, using appropriate protective equipment, and respecting both the fire hazards and regulatory requirements, you can safely work with this important material across its many applications.
Explore our Resource Library for more information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Non-Denatured Ethanol You Can Trust?
If you are looking for high-quality non-denatured ethanol for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's non-denatured ethanol is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.









