Isopropanol (Isopropyl Alcohol) Safety & Hazards: Essential Handling Guidelines
Isopropanol, also known as isopropyl alcohol, 2-propanol, or rubbing alcohol, is a colorless, flammable liquid with a characteristic alcohol-like odor widely used as a solvent, disinfectant, cleaning agent, and antiseptic. Isopropanol poses significant fire hazards, with a low flash point of approximately 12°C (53°F), and forms explosive vapor-air mixtures at concentrations between 2-12.7%. While moderately toxic if ingested and capable of causing central nervous system depression, skin and eye irritation, and respiratory tract irritation, isopropanol is generally safe when handled with proper precautions, including adequate ventilation, ignition source elimination, and appropriate personal protective equipment.
Professionals across healthcare, manufacturing, electronics, laboratories, and cleaning services rely on isopropanol daily. Understanding its safety profile helps you work confidently with this versatile chemical while avoiding the fire and health hazards it can create.
The material ignites easily, burning with flames so faint you might not see them in daylight. Vapors settle in low areas where they can travel unseen to distant sparks or flames. Swallowing isopropanol causes poisoning that affects your central nervous system, potentially leading to unconsciousness or worse. Yet with straightforward precautions, you can handle this chemical safely across countless applications from disinfecting surfaces to manufacturing electronics.


In this article, we’ll explore:
-
Chemical Hazards Overview
-
Exposure Risks & Health Effects
-
First Aid Measures
-
Handling & Storage Precautions
-
Spill & Leak Procedures
-
Fire & Explosion Hazards
-
Regulatory & Compliance Information
-
Common Formulations & Applications
-
Technical Documents & References
Chemical Hazards Overview
Isopropanol's primary danger is its flammability, though ingestion and high-vapor exposures also pose health risks worth understanding.
Hazard Classification
Under the Globally Harmonized System, isopropanol receives a flame pictogram (???? GHS02) for highly flammable liquid and vapor classified as Category 2. An exclamation mark (⚠️ GHS07) warns of serious eye irritation (Category 2A), skin irritation (Category 2), and the potential to cause drowsiness or dizziness from central nervous system effects (Category 3). The material is harmful if swallowed or inhaled and may be harmful if absorbed through the skin.
NFPA hazard ratings place isopropanol at Health: 1-2 (slight to moderate hazard), Flammability: 3 (serious fire hazard), and Reactivity: 0 (stable).
Primary Risks
Fire hazards dominate the risk profile. The flash point ranges from 12-18°C (53-64°F), meaning the liquid can ignite on a cool day if vapors contact an ignition source. Autoignition occurs at 399°C (750°F). The material forms explosive vapor-air mixtures between 2% and 12.7% concentration in air. Since vapors are heavier than air, they flow along floors and through ducts to reach ignition sources that may be far from where you're working.
Toxicity is moderate compared to many industrial solvents. The oral LD50 is approximately 5000 mg/kg in rats. High vapor concentrations cause central nervous system depression. Direct contact irritates eyes and respiratory passages. Swallowing creates an aspiration hazard if the liquid enters your lungs.
Exposure Risks & Health Effects
Knowing how isopropanol affects the body helps you recognize when exposures are occurring and respond before symptoms become severe.
Inhaling vapors irritates your respiratory tract, causing coughing and throat discomfort. High concentrations may trigger headache, dizziness, drowsiness, nausea, and central nervous system depression. Very heavy exposures can progress to narcotic effects, confusion, and unconsciousness. Normal handling at room temperature generally creates low hazard since isopropanol has moderate vapor pressure and doesn't readily fill the air with vapors unless spilled or heated.
Skin contact causes mild to moderate irritation with prolonged or repeated exposure. You may notice drying, cracking, and dermatitis developing over time. The material can be absorbed through the skin in significant amounts, though dermal absorption is generally considered less toxicologically significant than breathing vapors or swallowing the liquid. The defattening effect on skin with repeated exposure damages your skin's protective barrier.
Getting isopropanol in your eyes causes serious irritation with redness, tearing, pain, and blurred vision. Vapor exposure also irritates the eyes. Permanent damage is not expected with prompt treatment. Eye effects typically resolve with thorough rinsing.
Swallowing isopropanol is harmful and can be dangerous. The probable oral lethal dose in humans is approximately 240 mL, though as little as 20 mL has caused poisoning in some cases. Ingestion causes gastrointestinal irritation with nausea, vomiting, and diarrhea. Central nervous system depression may progress from initial excitement through headache, dizziness, drowsiness, and confusion to unconsciousness, coma, and possible death from respiratory failure. If you vomit after swallowing isopropanol, the liquid can enter your lungs, causing chemical pneumonitis, which can be fatal.
Repeated or prolonged exposure over time may damage your kidneys, liver, and central nervous system. Chronic inhalation may affect your respiratory system. Isopropanol is not classified as a carcinogen by ACGIH (A4), IARC (Category 3), NIOSH, NTP, or OSHA. Animal studies show no birth defects at doses toxic to the mother. Possible reproductive or developmental effects at very high exposures are based on limited animal data.
First Aid Measures
Quick, appropriate first aid response reduces harm from isopropanol exposure. The specific actions depend on how the contact occurred.
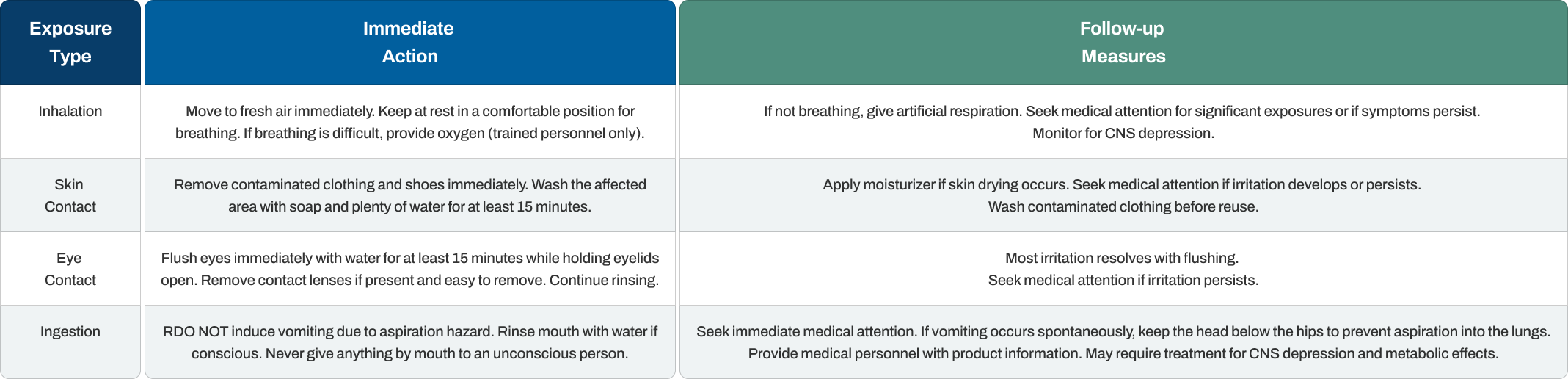
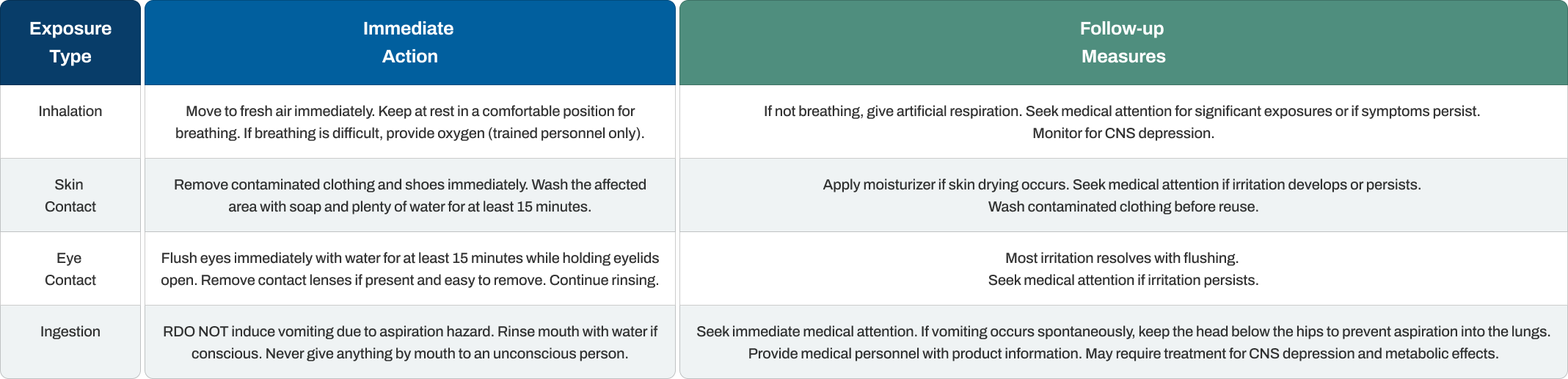
The aspiration hazard from swallowing isopropanol makes inducing vomiting dangerous. If the person vomits naturally, keeping their head lower than their hips helps prevent liquid from entering the lungs. Central nervous system depression can develop after ingestion, so medical observation is important even if the person seems fine initially.
Handling & Storage Precautions
Safe isopropanol handling centers on fire prevention while avoiding skin contact and vapor inhalation.
Safe Handling Procedures
Eliminate all ignition sources, including sparks, flames, hot surfaces, and static electricity, before opening containers. Use explosion-proof electrical, ventilating, and lighting equipment in areas where isopropanol vapors might accumulate. Ground and bond containers during transfer operations. Use non-sparking tools for opening containers and handling equipment.
Ensure adequate ventilation through general dilution or local exhaust systems. Wear chemical-resistant gloves (nitrile recommended), safety goggles, and protective clothing to prevent skin and eye contact. Avoid breathing vapors or mist. Wash hands thoroughly after handling and before eating, drinking, or smoking.
Storage Conditions
Store in cool, dry, well-ventilated areas away from heat, sparks, open flames, and hot surfaces. Keep containers tightly closed when not in use to prevent vapor release and evaporation. Use approved flammable liquid storage cabinets for quantities exceeding small amounts. Maintain storage temperature below 25°C (77°F) and protect from direct sunlight, which can heat containers and increase vapor pressure.
Keep away from incompatible materials. Do not pressurize, cut, weld, or expose containers to heat or flame. Inspect storage areas regularly for container damage, proper labeling, adequate ventilation, and absence of ignition sources.
Incompatible Substances
Keep isopropanol away from strong oxidizing agents, including peroxides, permanganates, nitrates, perchlorates, and chromium trioxide. Separate from strong acids and bases, halogens, and aluminum. Acetyl chloride reacts dangerously with isopropanol. Phosgene with iron salts creates hazardous reactions.
Isopropanol may form dangerously unstable peroxides with air or oxygen over time, particularly in partially filled containers with large headspace. The material is incompatible with 2-butanone (methyl ethyl ketone), which increases peroxide formation.
Spill & Leak Procedures
Isopropanol spills create immediate fire hazards requiring quick response to control vapors and eliminate ignition sources.
Emergency Response Steps
Eliminate all ignition sources immediately throughout the area. Evacuate unnecessary personnel and restrict access. Ensure adequate ventilation to disperse vapors, using explosion-proof fans if mechanical ventilation is needed.
Wear appropriate personal protective equipment, including chemical-resistant gloves, safety goggles, protective clothing, and respiratory protection for large spills. Use explosion-proof equipment only for all cleanup activities. Stay upwind and avoid low areas where vapors collect.
Containment Methods
Stop the leak if you can do so safely. Contain the spill with dikes or barriers to prevent spreading. Absorb with inert, non-combustible materials such as vermiculite, sand, or earth. DO NOT use combustible absorbents like sawdust or rags.
Prevent entry into waterways, sewers, basements, or confined spaces where vapors can accumulate and create explosion or toxic atmosphere hazards. Remember that vapors may travel considerable distances to reach ignition sources.
Proper Disposal
Recover spilled material where possible for reuse or proper disposal. Dispose of isopropanol and contaminated absorbents as hazardous waste according to federal, state, and local regulations. Consult a licensed hazardous waste disposal contractor for proper disposal methods.
Large quantities may require incineration in an approved facility with adequate emission controls. Do not discharge to drains or waterways. Isopropanol is subject to SARA Section 313 reporting requirements for releases.
Fire & Explosion Hazards
Fire represents the most serious immediate danger when working with isopropanol. Understanding its combustion behavior is essential for prevention and response.
Flammability
Isopropanol is a highly flammable liquid and vapor classified as DOT Class 3, Class IB flammable liquid. The flash point ranges from 12-18°C (53-64°F) in closed cup testing. Autoignition temperature is 399°C (750°F). Flammable limits span from 2.0% (lower explosive limit) to 12.7% (upper explosive limit) at 93°C.
Vapors are heavier than air and travel along the ground to distant ignition sources, where they can ignite and flash back to the spill. The material may form explosive vapor-air mixtures readily. Isopropanol burns with nearly invisible flames in daylight, similar to other alcohols.
This makes detecting fires difficult and increases injury risk. Over time, isopropanol may form dangerous peroxides with air, particularly in containers with significant headspace.
Extinguishing Media
Use alcohol-resistant foam (AFFF), dry chemical, carbon dioxide (CO2), or water spray or fog for extinguishing isopropanol fires. Water may be ineffective for actually extinguishing flames, but it should be used to cool containers and prevent fire spread.
Do NOT use straight water streams, which can spread burning liquid. For large fires, alcohol-resistant foam or water spray works best. For small fires, dry chemical, CO2, or dry sand are effective.
Firefighting Precautions
Firefighters should wear full protective gear, including a self-contained breathing apparatus and fire-resistant clothing. Approach fires from upwind to avoid smoke and vapor inhalation. Cool fire-exposed containers with water spray to prevent rupture from heat.
Be aware that vapors may travel to ignition sources and flash back to the fire area. Containers may explode when exposed to fire. Combustion produces toxic gases, including carbon monoxide and carbon dioxide. Vapors are heavier than air and collect in low areas, creating reignition hazards.
Regulatory & Compliance Information
Multiple agencies regulate isopropanol use, storage, and transportation based on its flammability and health effects.
OSHA Regulations
Isopropanol is regulated as a flammable liquid under 29 CFR 1910.106. OSHA's Permissible Exposure Limit (PEL) is 400 ppm (980 mg/m³) as an 8-hour time-weighted average, with a Short-Term Exposure Limit (STEL) of 500 ppm (1225 mg/m³) for 15 minutes. ACGIH recommends a Threshold Limit Value (TLV) of 200 ppm TWA with a 400 ppm STEL.
NIOSH's Recommended Exposure Limit (REL) is 400 ppm (980 mg/m³) for a 10-hour TWA and 500 ppm (1225 mg/m³) STEL. Employers must comply with the Hazard Communication Standard for labeling, safety data sheets, and worker training.
EPA & Environmental Impact
Isopropanol is not listed as a hazardous substance under CERCLA, so spills don't trigger the strict reporting requirements that apply to many chemicals. However, the material is subject to SARA Section 313 Toxics Release Inventory (TRI) reporting requirements if your facility uses threshold quantities. The chemical appears on state Right-to-Know lists in California, New Jersey, Pennsylvania, Minnesota, Massachusetts, and Florida.
In the environment, isopropanol is expected to biodegrade moderately in soil and water systems. The 96-hour LC50 for fish exceeds 100 mg/L, indicating low aquatic toxicity. Large releases may be dangerous to aquatic life at high concentrations.
DOT Transportation Regulations
Isopropanol falls under Hazard Class 3 (Flammable Liquid) for transportation. The proper shipping name is "Isopropanol" or "Isopropyl alcohol." The UN number is UN1219. Packing Group II classification reflects the moderate flash point.
Shipments require flammable liquid labels and placards on vehicles carrying bulk quantities. Passenger aircraft are limited to 5 L containers, while cargo aircraft can carry up to 60 L containers.
FDA/TTB Compliance
USP/NF grade isopropanol is available for pharmaceutical applications and must meet United States Pharmacopeia standards for purity and contaminants. The material is used in FDA-approved pharmaceutical formulations and as an antiseptic.
A 70% aqueous solution is sold as rubbing alcohol for topical antiseptic use in consumer and healthcare settings. Isopropanol is not approved for internal consumption or food contact applications.
Common Formulations & Applications
Different concentrations and grades of isopropanol serve specific purposes with varying safety considerations.
Anhydrous Isopropanol (99-100%): This highest purity form is used as a solvent in manufacturing, laboratory reagents, electronics cleaning, and pharmaceutical production. The lower flash point and higher flammability compared to diluted forms require the strictest fire safety precautions. Lab Alley offers anhydrous isopropanol in various sizes for industrial and laboratory applications.
Isopropyl Alcohol 70% (Rubbing Alcohol): The most common concentration for disinfection and antiseptic use provides optimal antimicrobial activity at 70% concentration. Lower flammability than the anhydrous form, but still flammable with a flash point of approximately 18-24°C. Widely used in healthcare, laboratories, and consumer products. Lab Alley supplies pharmaceutical and technical grade 70% isopropanol for diverse applications.
Isopropyl Alcohol 50-70%: Used in cleaning products, hand sanitizers, and general disinfection with moderate antimicrobial activity. Flammability and flash point vary by exact concentration. Suitable for general-purpose cleaning and disinfection applications across multiple industries.
Industrial Grades: Technical grade serves manufacturing needs, while denatured formulations meet specific industrial uses. Electronic grade isopropanol provides the high purity required for semiconductor and electronics manufacturing. Purity and specification requirements vary by application and industry standards. Lab Alley provides various industrial grades meeting different technical specifications.
Technical Documents & References
Accurate technical documentation ensures safe handling and regulatory compliance across different isopropanol applications.
Safety Data Sheets (SDS) provide concentration-specific safety information, including physical properties (flash point, vapor pressure, specific gravity), exposure limits, handling procedures, and emergency response information for different isopropanol grades and concentrations.
OSHA Chemical Sampling Information details exposure monitoring methods, PEL values, sampling procedures, and analytical methods for workplace air quality assessment to ensure compliance with exposure limits.
NIOSH Pocket Guide to Chemical Hazards provides exposure limits, physical properties, chemical incompatibilities, measurement methods, and personal protective equipment recommendations based on comprehensive industrial hygiene research.
Certificate of Analysis (COA) verifies purity (typically 99% or 70%), water content, specific gravity, absence of contaminants, and compliance with applicable standards (USP, ACS reagent grade, technical grade, or electronic grade specifications) for each batch.
Final Note
Isopropanol serves countless essential functions across healthcare, manufacturing, electronics, and cleaning applications. Its effectiveness as a solvent and disinfectant makes it indispensable in modern operations. Success with isopropanol safety comes from respecting its flammability while managing the more moderate health hazards it presents.
Consistent fire prevention through ignition source elimination, proper ventilation to control vapor concentrations, and appropriate personal protective equipment to prevent skin contact and inhalation form the foundation of safe isopropanol handling. Understanding how concentration affects both antimicrobial effectiveness and flammability helps you select the right product for each application.
Lab Alley provides high-quality isopropanol in multiple grades and concentrations to meet your specific needs, whether you're disinfecting medical equipment, cleaning electronic components, or using it as a manufacturing solvent. By following the safety protocols outlined in this guide and maintaining awareness of fire hazards, you can work confidently with this versatile chemical across your operations.
Explore our Resource Library for more information.
Our Customer Care team is also available for more information and documentation, including chemical Safety Data Sheets.
Need Isopropanol You Can Trust?
If you are looking for high-quality isopropanol for your lab, product line, or research facility, be sure to visit our store to explore our trusted selection and request a quote.
As your trusted partner in chemical supply, Lab Alley is always striving to meet the growing demands of our customers. We want you to be able to access all of your chemical needs in one place, along with streamlined online ordering and fast shipping.
Due to popular demand from our valued clientele, Lab Alley's isopropanol is available in a range of purities and packaging options to suit various commercial, laboratory, and personal needs.














