About Chloroform
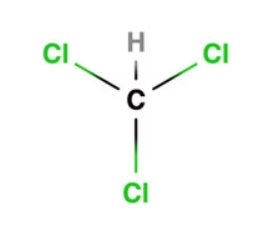
Chloroform (IUPAC name, trichloromethane) has the formula CHCl3, and it is a clear, colorless, strong-smelling, and dense liquid. It is a widely used industrial solvent, and has anesthetic and sedative properties, although its specific use as such in medicine has long been discontinued since it is toxic to the liver and kidneys in humans.
Some chloroform occurs naturally from fungi and seaweed. It is used as a reagent in the production of several important chemicals such as PTFE (Teflon) and historically (since the use of many of these compounds are now strictly regulated) chlorinated and fluorinated refrigerants. Chloroform can be released into the environment as a by-product of the chlorination of water, and of bleaching in the paper and pulp industry.
Chloroform is used as an extraction solvent for fats, oils, greases, rubber, waxes, gutta-percha, resins, lacquers, floor polishes, artificial silk manufacture, gums and adhesives. It is utilized as an industrial solvent in the extraction and purification of some antibiotics, alkaloids, vitamins and flavors.
Common uses of Chloroform
- Eextraction solvent
- As a solvent in NMR Spectroscopy
- As a precursor to the production of Teflon (PTFE)
- Historically as a precursor to the production of chlorofluorocarbon refrigerants
- Historically as an anesthetic
Industries
- Chemical Industry
- Pesticide Industry
- Rubber Industry








