Hydrochloric Acid
Pure Hydrochloric Acid (HCl) is an inorganic acid. It is also known as Hydrochloric Acid and Aqueous Hydrogen Chloride. It is a clear, colorless to light yellow liquid at room temperature with a potentially irritating odor. It has an approximate Ka of 1.3 x 10-6, and as such Hydrochloric Acid is a strong acid.
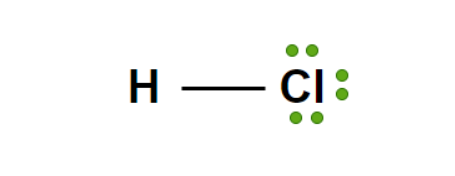
Hydrochloric Acid has a wide range of uses including as a lab reagent, in the manufacture of polyvinyl chloride (PVC), as a cleaning agent especially on masonry, in 'steel pickling', a process to clean and purify steel, and in pH control in both industrial and home use such as the regulation of the acidity of swimming pools. It is a 'workhorse' reagent in the chemical industry with a multitude of uses.
Industries that use Hydrochloric Acid
- Textiles
- Pharmaceutical & Nutraceutical
- Chemical
- Metal processing
- Food & Beverage
Common Uses and Applications for Hydrochloric Acid
- Regent in the chemistry lab
- Industrial cleaning
- Precursor in chemical manufacture
- Metal processing (steel pickling)
- Controlling pH in domestic and industrial situations




























