About Dimethylformamide
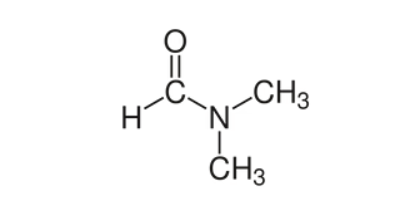
Dimethylformamide (IUPAC name N,N-dimethylfomamide) is structurally the same as formamide but with two methyl groups replacing the two hydrogens. It is a clear and odorless liquid, although impurities of dimethylamine may result in a fishy aroma. It is prepared via the reaction of dimethyamine with either methyl formate or carbon monoxide. It is miscible with water, and has a wide range of applications as a solvent.
Dimethylformamide is often used as a solvent or reagent in synthetic reactions, including aldehyde synthesis, acyl halide synthesis, and in the Heck reaction. The pharmaceutical industry uses Dimethylformamide as a solvent for peptide coupling reactions. It has been used to recover olefins via extractive distillation. It is also used in the manufacturing process of adhesives, fibers, and synthetic leathers.
Common Uses and Applications
- Peptide-coupling in pharmaceuticals
- Pesticide production
- Organic synthesis
- Solid-phase peptide synthesis
- NMR spectroscopy standard
- Adhesives
- Fibers and synthetic leathers
Industries
- Research Laboratories
- Pharmaceutical Industry
- Textiles industry








